GCSE/IGCSE Organic Chemistry - Part 3 - Alkenes
Introduction to Alkynes
Overview of Organic Molecules
- The video introduces the topic of alkynes, part of an organic chemistry series, following a previous discussion on alkanes.
- Alkenes are defined as hydrocarbons containing carbon-carbon double bonds, distinguishing them from alkanes which only have single bonds.
Key Characteristics of Alkynes
- An alkyne is characterized by at least one carbon-carbon triple bond, making it unsaturated and reactive due to the presence of this bond.
- The double bond in alkenes serves as a functional group where most reactions occur, leading to greater reactivity compared to alkanes.
General Formula and Structure
Understanding Alkenes
- The general formula for an alkene with one double bond is CnH2n, indicating that there are twice as many hydrogen atoms as carbon atoms.
Branched Alkenes
- Alkenes can exist in branched forms; for example, a molecule with four carbon atoms and a branch results in names like methyl butene.
Reactions of Alkenes
Types of Reactions
- Alkenes can undergo various reactions: addition (hydrogenation), hydration, combustion, and polymerization.
Hydrogenation Process
- In hydrogenation, the carbon double bond breaks to allow hydrogen atoms to join each carbon atom, converting an alkene into an alkane.
Saturation Concept
- This process also illustrates how unsaturated alkenes become saturated when they react with hydrogen.
Addition Reactions
Addition of Halogens
- Similar to hydrogenation, halogen addition involves breaking the double bond in an alkene and forming a halogenated product (halogen alkane).
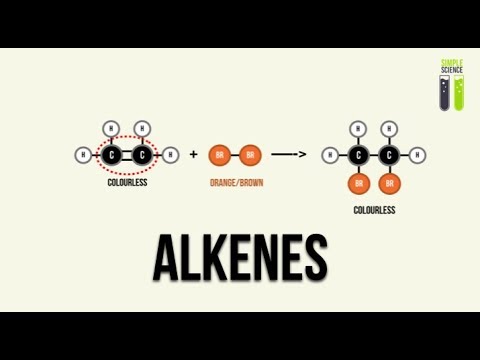
Visual Test for Alkenes
- A practical test for detecting alkenes involves adding bromine water; if decolorization occurs (brown liquid turns colorless), it indicates the presence of an alkene.
Hydration Reaction
Formation of Alcohol
- Hydration involves reacting steam with an alkene. The reaction breaks the double bond and forms alcohol through bonding between water's components and the carbons in the alkene.
Understanding Alkene Reactions and Polymerization
Formation of Alcohol from Alkenes
- The reaction to form an alcohol from an alkene requires specific conditions, including a pressure of 60 bar and the use of phosphoric acid as a catalyst.
- It is essential to remember these conditions for successful alcohol synthesis from alkenes.
Introduction to Polymerization
- Polymerization occurs when the double bonds in alkenes break, allowing them to join with other alkene molecules, resulting in long-chain molecules known as polymers.
- This process transforms single substances into longer molecular structures under certain conditions.
Key Concepts in Polymer Chemistry
- A monomer serves as the building block for polymers; it is the alkene whose carbon double bonds are broken during polymerization.
- Each polymer can consist of up to 20,000 repeating units, which represent individual segments of the entire polymer chain.
Reaction Conditions for Polymerization
- Typical polymerization reactions occur at high temperatures (around 200 degrees Celsius) and pressures (approximately 2,000 bar), particularly for polyethylene.
- Memorizing these reaction conditions is crucial for understanding how polymers are formed effectively.