¿Qué son los FULLERENOS? 🧪
What are Fullerenes?
Introduction to Fullerenes
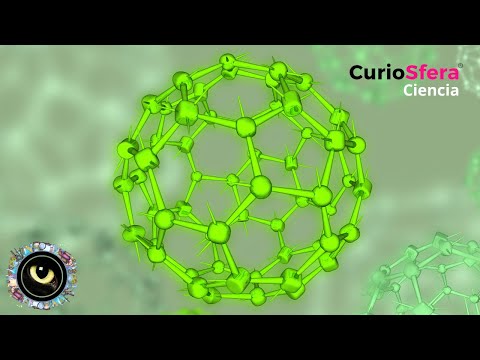
- Fullerenes are new carbon molecular structures, recognized as the third stable molecular form of carbon after diamond and graphite.
- The patent specification refers to the first method for producing these substances, which was quickly replaced, not indicating the discovery itself.
Discovery Process
- In 1985, an experiment at Rice University involved Harold Kroto, Richard Smalley, and Robert Curl aiming to simulate conditions near a star's surface to observe large molecule formation in space.
- They used a laser on a carbon surface in helium presence; gaseous carbon combined with helium formed clusters. Initial tests included hydrogen and nitrogen but were later limited to nitrogen.
- Cooling the gas revealed that C60 (60 carbon atoms in one molecule) was the most abundant cluster observed. This structure resembled Buckminster Fuller’s geodesic dome from Expo 67.
Naming and Structure
- Unable to recreate it digitally, they resorted to paper models and named it "buckminsterfullerene," later shortened to "fullerenes." An alternative name considered was "soccerene."
Applications of Fullerenes
- Carbon atoms in fullerenes bond well with others, forming long chains called polymers used in products like plastic cups and bottles.
- A unique property of fullerenes is that some electrons are "delocalized," allowing easy addition of other atoms for applications such as single-crystal superconductors or insulators.
Future Prospects
- By 1991, interest surged with numerous reports on fullerenes emerging. A promising idea involves altering their structure into buckytubes—hollow fibers significantly stronger than steel—potentially useful for drug delivery systems or radiation shields.