CONFIGURACION ELECTRONICA (paso a paso y fácil)
New Section
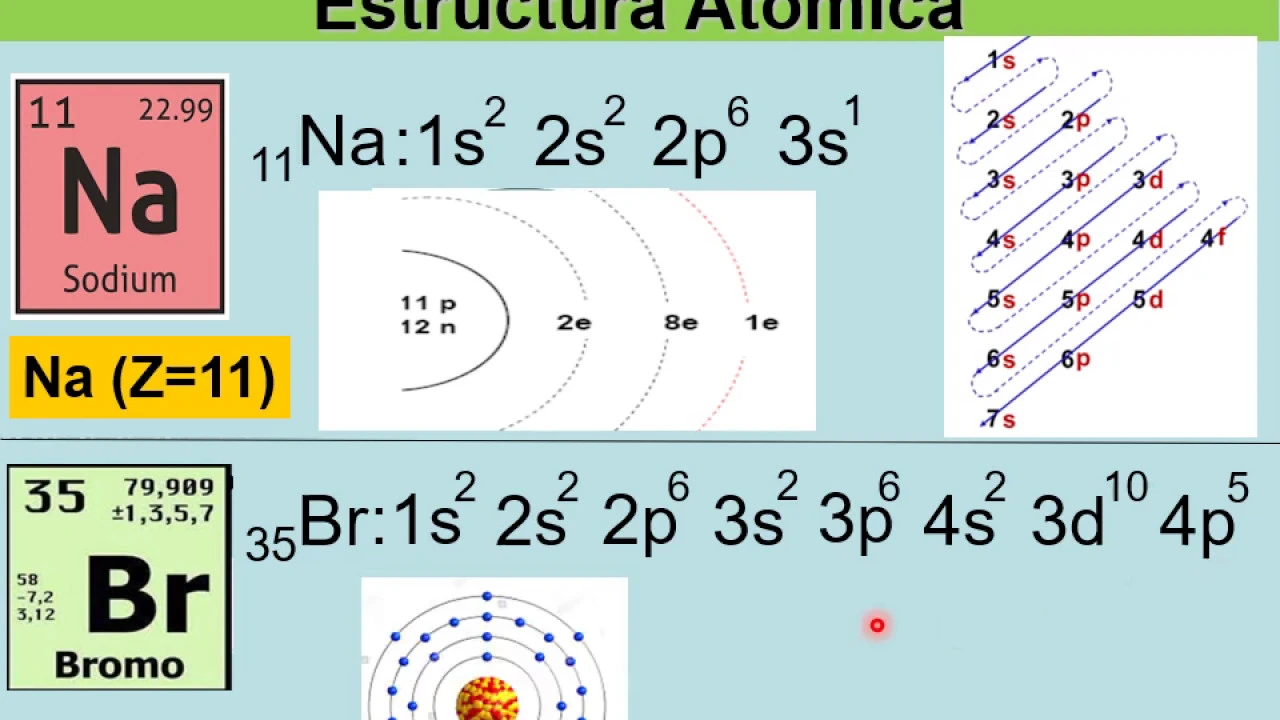
In this section, the speaker introduces the topic of electronic configuration by discussing the structure of an atom, emphasizing the arrangement of protons, neutrons, and electrons in different energy levels.
Introduction to Electronic Configuration
- The atom consists of a nucleus containing protons and neutrons, with electrons located in energy levels.
- Electrons are distributed across energy levels denoted by letters s, p, d, or f.
- Electronic configuration is represented using quantum notation indicating energy level number, sublevel letter (s,p,d,f), and electron count.
New Section
This part delves into practical examples of electronic configurations using elements like helium. It also introduces the concept of atomic number and its significance in determining electron count.
Practical Examples and Atomic Number Significance
- Helium's atomic number is 2, representing both protons and electrons.
- Atomic number indicates the electron count in an atom.
- Utilizing a diagonal rule diagram aids in determining electronic configurations efficiently.
New Section
Here, a detailed explanation is provided on how to construct a diagonal rule diagram for electronic configurations based on specific columns representing different energy levels.
Constructing Diagonal Rule Diagram
- Create columns from 1 to 7 for different energy levels.
- Label each column with s,p,d,f designations.
- Use a diagonal line pattern to represent electron distribution following arrow directions.
New Section
This segment focuses on understanding electron capacity per sublevel (e.g., s,p,d,f), guiding the order of filling electrons during electronic configuration determination.
Electron Capacity and Filling Order
- Sublevels have maximum electron capacities: s(2), p(6), d(10), f(14).
- Following arrow directions in the diagonal diagram determines electronic configuration order comprehensively.
New Section
The final part demonstrates practical application through exercises involving determining electronic configurations for oxygen and magnesium based on their atomic numbers.
Practical Application: Oxygen and Magnesium Configurations
- Exercise involves calculating electronic configurations for oxygen and magnesium given their atomic numbers.
Configurations and Structures of Atoms
In this section, the speaker discusses the configuration and structure of atoms, focusing on elements like oxygen and magnesium. The relationship between electron configurations and atomic structures is explored through detailed examples.
Understanding Electron Configurations
- Magnesium with an atomic number of 12 has 12 electrons, initiating its electron configuration process.
- For oxygen, with atomic number 8, the electron configuration is determined by filling up energy levels systematically.
- Examining the electron configurations of oxygen and magnesium reveals a close connection between electronic configurations and atomic structures.
Relating Electron Configurations to Atomic Structure
- By calculating the electrons placed in each level for oxygen, a clear picture of its atom's structure emerges.
- Understanding how electrons are distributed across different energy levels provides insights into the overall atom structure.
Complex Atom Configuration: Bromine Example
This part delves into a more intricate example by analyzing the electron configuration of bromine (atomic number 35). The process involves detailing how electrons fill various orbitals to form the atom's structure.
Analyzing Bromine's Electron Configuration
- Starting with bromine's atomic number 35, the speaker illustrates how electrons occupy different sublevels following specific rules.
- Calculating the total electrons in each orbital for bromine aids in visualizing its atom's arrangement comprehensively.
Visualizing Bromine's Atomic Structure
- By summing up electrons in each level for bromine, a complete depiction of its atom's structure is achieved.