Estructura y unión de carbohidratos | Enlaces Glucosídicos
Understanding Carbohydrates: Structure and Function
Introduction to Carbohydrates
- The video begins with a recap of the previous discussion on carbohydrates, focusing on their characteristics and functions.
- It emphasizes the importance of understanding carbohydrate structures to differentiate between various types.
Monosaccharides: The Building Blocks
- Monosaccharides are defined as the simplest form of carbohydrates, consisting of a central chain of carbon atoms connected by single bonds.
- Key functional groups in monosaccharides include aldehyde or ketone groups, which influence their properties; aldehydes are at one end while ketones are in the middle.
- Hydroxyl groups (–OH) attached to carbon atoms make carbohydrates hydrophilic, explaining why substances like table sugar dissolve easily in water.
Structural Variations Among Monosaccharides
- Differences among monosaccharides can arise from variations in the number of carbon atoms; for example, ribose has five carbons (pentose), while glucose has six (hexose).
- Functional group differences also exist; glucose is an aldose (aldehyde group), whereas fructose is a ketose (ketone group), leading to distinct properties.
Isomers and Their Importance
- Isomers have the same molecular formula but differ structurally. For instance, galactose is an isomer of glucose due to different arrangements around one carbon atom.
- This structural variation results in differing physical properties despite identical formulas.
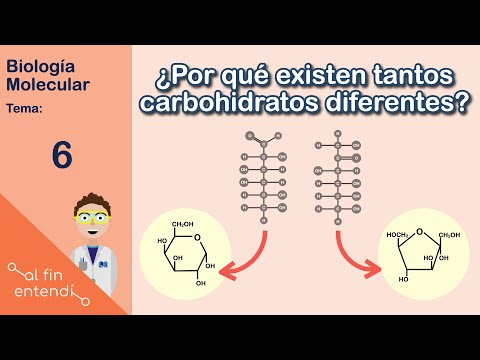
Cyclic Structures and Stability
- Monosaccharides often form cyclic structures when dissolved in water, enhancing stability compared to linear forms.
- Common cyclic forms include hexagons and pentagons; these shapes allow for more stable interactions with surrounding water molecules.
Formation of Disaccharides
Formation of Disaccharides and Polysaccharides
Understanding Glycosidic Bonds
- A hydrogen atom is removed from one glucose molecule, allowing it to bond with another glucose molecule through an oxygen bridge, forming a glycosidic bond. This results in the creation of maltose, a disaccharide.
- The monosaccharides involved in glycosidic bonding do not have to be identical; for instance, fructose can bond with glucose to form sucrose, commonly known as table sugar.
Role of Glucose in Energy Storage
- When we consume more glucose than needed for immediate energy, the excess can be stored in the liver or muscles as glycogen. This polysaccharide serves as a long-term storage form of glucose.
- Glycogen consists of long chains of glucose molecules that can branch at various points by linking through different carbon atoms. The fundamental principle remains consistent: they are joined via glycosidic bonds facilitated by hydroxyl (OH) groups.
Summary Insights on Polysaccharide Formation