Cours N°4 - Force électrique et loi de Coulomb
Introduction to Electric Force
Course Overview and Context
- The speaker introduces the continuation of an electronics course, acknowledging a delay in content delivery due to recent personal commitments.
- Expresses gratitude for the growing audience and hints at future plans for the channel in 2017.
Recap of Previous Lessons
- The lesson focuses on electric force, building upon previous discussions about free electrons and charge types.
- A reminder that free electrons are the fundamental charge used in electronics, with both positive and negative charges being relevant.
Understanding Charge Movement
Key Concepts of Electronics
- Defines electronics as the controlled movement of charges, emphasizing understanding what a charge is and how to manipulate it.
- Assumes prior knowledge about charges from previous lessons, transitioning into how to move these charges effectively.
Electric Force Explained
Fundamental Principles
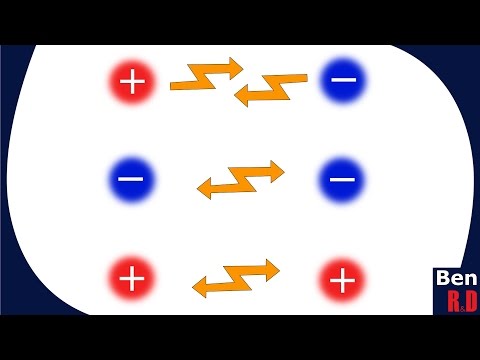
- Introduces electric force as a key concept where opposite charges attract while like charges repel each other.
- Clarifies that this attraction/repulsion phenomenon is part of electromagnetism, which encompasses various physical phenomena.
Application of Electric Force
- Discusses how electric force can assist in moving charges within electronic systems.
- Proposes an analogy involving a car (representing an electronic system), illustrating challenges faced without a 'motor' for charge movement.
Practical Implications of Electric Force
Moving Charges with Electric Forces
- Revisits the car analogy by introducing an electron as a charge needing movement; emphasizes using electric forces for this purpose.
- Explains that placing a positive charge near an electron will attract it, demonstrating practical applications of electric force in moving charges effectively.
Understanding Electric Force
Introduction to Electric Force
- The electric force is a practical concept that can be demonstrated physically, contrasting with more abstract theories. The speaker emphasizes the importance of observable phenomena in understanding this force.
Observing Electric Force
- The speaker plans to demonstrate the electric force using charged objects, highlighting that it is not merely theoretical but a tangible aspect of physics.
Charging Objects through Friction
- A simple method for charging objects involves friction; rubbing two items together can transfer charge and electrify them.
- Common experiences, such as getting shocked after walking barefoot and touching metal, illustrate how friction leads to charge accumulation on the body.
Practical Examples of Electrification
- When walking barefoot, friction with the ground transfers charge to the body, leading to a small shock when touching conductive materials like doorknobs or radiators.
- Everyday items like a ruler and calculator can be used for demonstration, although they may not be ideal for observing electric forces compared to other materials.
Effective Materials for Demonstration
- Using a plastic straw and an old cloth is suggested as an effective combination for demonstrating electrification through friction.
- Rubbing these two items together will result in electron transfer from the cloth to the straw, creating charged objects suitable for observation.
Explaining Charge Transfer Mechanism
Simplified Explanation of Electrification
- The speaker aims to explain what happens at a molecular level during electrification while acknowledging that this explanation will be simplified and may overlook complexities.
Conceptualizing Matter in Terms of Potential Wells
- Both the cloth and straw are described as consisting of atoms arranged in potential wells. This analogy helps visualize how charges interact during friction.
Differences Between Materials
- The atomic structure of each material differs; thus, their potential wells vary in depth. This difference plays a crucial role in charge transfer during contact.
Energy Barriers During Charge Transfer
- A potential energy barrier forms between the two materials due to differences in their atomic structures. This barrier influences how easily electrons can move from one object to another.
By structuring these notes chronologically with timestamps linked directly back to specific parts of the transcript, readers can efficiently navigate through key concepts related to electric force and its practical demonstrations.
Understanding Electron Transfer Through Friction
The Basics of Electron Movement
- The discussion begins with a simplification of complex phenomena to understand electron transfer through friction.
- When two objects are rubbed together, energy is introduced at the friction point, allowing an electron from the cloth to potentially be released.
- However, the initial energy applied is insufficient for the electron to overcome the potential barrier and move to the straw's energy level.
Increasing Energy for Electron Transfer
- By applying more vigorous friction between the straw and cloth, additional energy is provided, enabling the electron to gain enough energy to surpass the potential barrier.
- An important note is that electrons are not simple particles; they exist as negative charges within specific energy levels. Sufficient friction can free an electron from its potential well in the cloth.
Consequences of Stopping Friction
- Once sufficient friction has been applied and an electron is freed, stopping this action causes it to lose acquired energy due to its nature as a charge spread over an energy layer.
- As it loses energy, it will fall back into the first potential well it encounters—in this case, that of the straw—resulting in heat loss during this process.
Summary of Electron Transfer Process
- The process involves rubbing straw against cloth which introduces enough energy to release an electron from its potential well in the cloth.
- If rubbed harder, more electrons can be freed and elevated above both materials until friction stops and they fall back into a lower potential state.
Implications of Charge Creation
- This transfer results in a positive charge (due to lost electrons creating holes) on the cloth while simultaneously imparting a negative charge on the straw due to gained electrons.
- Thus, one object becomes positively charged while another becomes negatively charged; their absolute charges are equal but opposite in sign.
Quantifying Charge Transfer
- Rubbing these materials typically generates several tens of nanocoulombs of charge—equivalent to hundreds of billions of electrons being transferred.
- It’s emphasized that this explanation simplifies a much more complex reality regarding electrification through friction.
Understanding Electric Forces and Charge Interactions
Introduction to Electric Forces
- The phenomenon of electric force explains how two oppositely charged objects attract each other, suggesting that a cloth should attract a straw.
- An experiment is set up to verify this attraction using a lightweight fishing line to suspend the straw above a lamp.
Conducting the Experiment
- The straw is suspended to allow for observation of weak electric forces; even slight movements can indicate attraction when the cloth is rubbed against it.
- After rubbing the cloth on the straw, the expectation is that they will attract due to their opposite charges.
Observing Attraction
- Upon stabilization, it becomes evident that the cloth attracts the straw, confirming the initial hypothesis about electric forces.
- A closer look reveals clear attraction between both objects, demonstrating that opposite charges indeed exert an attractive force on one another.
Testing Repulsion Between Like Charges
- The next step involves testing whether like charges repel each other by electrifying a second straw with negative charge.
- Two negatively charged straws are expected to repel each other based on established principles of electrostatics.
Results of Repulsion Experiment
- After charging both straws, they are observed not touching but repelling each other effectively, showcasing action at a distance due to electric forces.
- This clear demonstration reinforces understanding of electric forces acting between like charges and confirms their repulsive nature.
Conclusion and Implications for Electronics
- The experiments illustrate fundamental concepts in electronics: moving charges under controlled conditions. However, true electronic control requires more than just movement; it necessitates understanding these forces quantitatively.
- To advance in electronics, it's essential to quantify electric force—understanding how much force acts between charges based on their signs and distances apart.
Quantifying Electric Force
- It’s noted that electric force must be expressed in Newton (N), linking back to Isaac Newton's contributions in classical mechanics.
- Understanding units and magnitudes helps contextualize everyday experiences with gravity and related forces.
Understanding Electric Forces and Coulomb's Law
Introduction to Newton and Electric Force
- The concept of a Newton is introduced as the force that provides an acceleration of 1 meter per second squared to a mass of 1 kg, giving context to the scale of electric forces.
Exploring Electric Charges
- A question arises regarding the force between two electric charges, Q1 and Q2, prompting an exploration into quantifying this force, referred to as FE.
Experimental Approach
- The speaker suggests using experimental methods similar to previous examples by varying parameters such as charge values (Q1, Q2) and distance between them to measure electric forces.
Historical Context: Charles Augustin Coulomb
- Reference is made to Charles Augustin Coulomb, who conducted foundational work on electric forces over 200 years ago. His contributions led to naming the unit of electric charge after him.
Coulomb's Law Explained
- Coulomb discovered that the electric force (FE) between two charges (Q1 and Q2) can be expressed mathematically as FE = K cdot Q1 cdot Q2/r^2 , where K is a constant dependent on the medium.
Understanding Mathematical Implications
- The significance of mathematical equations in physics is emphasized; they represent concrete relationships rather than abstract concepts.
Proportionality in Electric Force
- It’s noted that the electric force is directly proportional to the magnitudes of charges Q1 and Q2. Larger charges result in stronger forces. Additionally, FE depends on K, which varies with different media.
Distance Factor in Electric Force
- The equation indicates that the electric force decreases with increasing distance (r), specifically inversely proportional to r^2 . This relationship suggests that greater separation leads to weaker forces.
Conceptualizing Influence Between Charges
- A simplified model illustrates how charge Q1 emits an influence received by charge Q2. This influence spreads uniformly in all directions from Q1.
Divergence of Influence Over Distance
- The emitted influence from charge Q1 dilutes over a larger surface area as it expands spherically. This phenomenon explains why strength diminishes with distance.
Surface Area Relation
- As radius increases, so does surface area; thus, influence diminishes not just with distance but also due to spreading across a larger sphere's surface area.
Key Insight: Inverse Square Law
- It’s clarified that while electrical force relates inversely with distance R, it actually correlates inversely with the surface area of a sphere defined by radius R—highlighting why r^2 appears in Coulomb's law instead of just r alone.
Conclusion on Mathematical Representation
- A brief discussion concludes about how constants like K account for variations across different mediums while maintaining consistency within Coulomb's law framework.
Understanding Coulomb's Law and the Constant K
The Role of 4π in Coulomb's Equation
- The constant 4π is integrated into the constant K for mathematical simplicity, even though it does not explicitly appear in the equation.
- Coulomb's law states that the electric force between two charges (Q1 and Q2) is proportional to their magnitudes and inversely proportional to the square of the distance (R²) between them.
Issues with Constant K
- There are ambiguities regarding constant K; it does not solely represent the medium since it also incorporates 4π, indicating a lack of clarity in its physical meaning.
- Historically, Coulomb did not fully understand what his constant represented due to limitations in scientific knowledge at that time, particularly lacking access to modern atomic models.
Understanding Permittivity
- To grasp constant K better, it's essential to recognize that it includes an inverse proportionality related to 4π and needs a parameter representing the medium affecting electric force.
- The key parameter describing how electric force varies with different media is called permittivity (ε), which indicates how material affects electric interactions.
Physical Implications of Permittivity
- Permittivity is linked to fundamental physical phenomena occurring within materials, varying from one substance to another based on their properties.
- It’s crucial to remember that permittivity expresses how electric forces between charges depend on their surrounding medium.
Understanding Electric Force and Permittivity
The Nature of Electric Force
- The electric force between two charges (Q1 and Q2) is inversely proportional to the permittivity of the medium in which they are placed.
Constant K and Its Components
- The constant K incorporates an inverse proportionality related to both 4π and ε, leading to the formulation k = 1/4pi epsilon .
Coulomb's Law Equation
- Substituting K into Coulomb's law results in the equation f = k cdot q_1 q_2/r^2 , where k = 1/4pi epsilon .
Permittivity Values in Electronics
- A table of common permittivity values for materials used in electronics is provided; permittivity has units that are the inverse of those for K.
Key Value: Vacuum Permittivity
- The vacuum permittivity, denoted as ε₀, is a reference value at approximately 8.85 times 10^-12 C^2/(N·m^2) .
Air vs. Vacuum Permittivity
- The permittivity of dry air is nearly identical to that of a vacuum, allowing simplifications in calculations by using ε₀.
Variability in Ceramic Materials
- Ceramics exhibit a wide range of permittivities (50 to 10,000), making them versatile materials in electronic applications.
Limitations of Coulomb's Law
Scalar Nature of Forces
- Coulomb's law provides only a scalar value for force without direction; this presents limitations since forces also have directional components.
Understanding Scalars vs. Vectors
- A scalar represents only magnitude (a number), while vectors include both magnitude and direction—essential for accurately describing forces.
Importance of Directionality
- For example, the electric force on charge Q1 due to Q2 not only has a magnitude but also points from Q1 towards Q2.
Vector Representation of Electric Force
Defining Vectors
- A vector expresses both magnitude (length) and direction (indicated by an arrow), essential for representing physical quantities like force accurately.
Application to Electric Forces
- In scenarios with like charges repelling each other, vector representation effectively conveys both the strength and directionality of the electric force acting on each charge.
Understanding Electric Force as a Vector
Introduction to Electric Force
- The discussion begins with the need to express electric force in vector form, specifically focusing on the force exerted on charge Q1 within a system of charges Q1 and Q2.
- The scalar value of the electric force is known from Coulomb's law, represented as F = K Q_1 Q_2/R^2 , where K is Coulomb's constant.
Direction of Electric Force
- To determine the direction of the electric force, a mathematical reminder is provided about multiplying a vector by a scalar, resulting in a new vector that maintains direction but alters magnitude.
- A unit vector is introduced, which has a magnitude of one (one Newton in this context) and points in the desired direction—specifically from charge 2Q2 towards charge Q1.
Formulating the Electric Force Vector
- By multiplying the known scalar value of electric force by this unit vector, we can derive the actual electric force vector acting on charge Q1.
- This multiplication ensures that both direction and magnitude are accurately represented in our final expression for electric force.
Summary of Key Concepts
- The conclusion emphasizes that the electric force vector can be expressed as vecF_E = F_E cdot vecu , where F_E is the scalar value and vecu is the unit vector indicating direction.
- The formula for Coulomb's law in its vector form becomes vecF_E = KQ_1Q_2/R^2cdotvecu , providing clarity on how to represent forces between charged particles.
Importance of Understanding Electric Forces
- It’s noted that while detailed mathematical understanding may not be necessary for basic electronics, grasping what these equations signify is crucial for practical applications.
- Acknowledgment that many learners may have encountered Coulomb's law previously but might not fully understand its implications or historical context.
Critique of Traditional Teaching Methods
- A critique arises regarding how Coulomb's equation has been taught historically; it suggests that students often learn an outdated version without understanding its relevance today.
- Emphasis on retaining meaningful interpretations rather than rote memorization of constants like K, which may seem arbitrary or confusing without proper context.
Understanding Coulomb's Law
Introduction to Coulomb's Law
- The formula for the electric force F is expressed as F = k cdot Q_1 cdot Q_2/4piepsilon r^2 , where k is a constant, and it mirrors the equation taught in school.
- The speaker emphasizes that this form of Coulomb's law is beneficial because it provides comprehensive insight into electric force, despite being more complex than simpler versions.
Key Components of the Equation
- Understanding each term in the equation is crucial; even without grasping the mathematical derivation, knowing what each component signifies is essential for practical applications in electronics.
Electric Force and Permittivity
- The electric force F_E acting on charge Q_1 depends inversely on the permittivity epsilon of the medium surrounding charges.
- This means that higher permittivity results in weaker electric forces between charges.
Charge Interaction
- The strength of the electric force increases with larger values of charges Q_1 and Q_2 ; thus, greater charge magnitudes lead to stronger interactions.
Distance Effect on Electric Force
- The term 1/(4pi r^2) indicates that as distance r between charges increases, the magnitude of electric force diminishes spherically.
- It’s important to note that this divergence implies that moving away from a charge reduces its influence significantly.
Directionality of Electric Force
- The unit vector in the equation indicates that electric force has a specific direction; for instance, when analyzing forces on charge Q_1, it points towards charge Q_2.
Practical Implications in Electronics
- Understanding these principles is vital for practical electronics applications. Recognizing how electric forces operate will aid comprehension when working with circuits.
Exploring Basic Electronic Circuits
Circuit Overview
- A simple electronic circuit consisting of a battery and a light bulb serves as an example to illustrate how electrical forces manifest practically within components.
Chemical Energy Conversion
- In batteries, chemical energy conversion leads to electron release, creating free electron pairs and holes. This process results in slight positive and negative charges accumulating at different terminals.
Conclusion
The discussion highlights fundamental concepts related to Coulomb's law and its implications for understanding basic electronic circuits.
Understanding Electric Forces in a Battery
Attraction Between Charges
- The electric force between opposite charges causes them to attract each other, meaning holes are drawn towards free electrons and vice versa.
Purpose of Free Electrons
- Free electrons exist primarily to fill the holes, which is clarified by the electric force acting on them. This relationship highlights their role in maintaining charge balance within the system.
Paradox of Constant Charge
- Despite being subjected to an attractive force from free electrons, the positive charge remains constant and stationary, indicating a paradox that needs resolution.
Balancing Forces in a Battery
- The stability of charges implies there must be another force counteracting the electric force; this opposing force is equal in magnitude but opposite in direction. This leads to the concept of electromotive force (emf).
Role of Electromotive Force
- The battery generates an electromotive force (emf) that maintains a constant potential difference across its terminals, essential for its function as a generator. Emf is crucial for sustaining energy output through chemical reactions within the battery.
Energy Production and Work
- A chemical reaction within the battery releases energy, which can be interpreted as work done by providing a constant emf that separates charges until equilibrium with electric forces is achieved. This process illustrates how batteries convert chemical energy into electrical work.
Implications Beyond the Battery
- Understanding these principles sets up further exploration into how these forces operate outside of the battery context, particularly regarding electron movement in circuits. This transition indicates deeper insights into electronic systems will follow.
Hypothetical Scenario Setup
- To explore electron behavior further, a hypothetical scenario is introduced where a stationary battery exists in a vacuum with an escaped free electron nearby; this setup aims to analyze interactions without external influences complicating observations.
Importance of Attention
- Emphasis is placed on understanding these concepts as they are foundational for grasping why free electrons move within electronic circuits; thus, active engagement during this explanation is encouraged for better comprehension.
Understanding the Movement of Free Electrons in a Circuit
The Setup: Charges and Forces
- The discussion begins with the scenario outside a battery, focusing on three charges: a free electron, the negative terminal charge, and the positive terminal charge of the battery.
- The primary interest is in what happens to the free electron within this system of charges, indicating that electric forces will be at play.
- The free electron experiences two distinct electric forces: one from the negative terminal (repulsion) and another from the positive terminal (attraction).
Analyzing Electric Forces on the Electron
- The first force acting on the free electron is repulsive due to its proximity to the negative terminal; this force is significant because of their like charges.
- Conversely, there is an attractive force exerted by the positive terminal. This force is weaker since it acts over a greater distance compared to that from the negative terminal.
- As a result of these two forces, there exists a resultant force directing movement towards the positive terminal, causing motion in that direction.
Continuous Motion and Force Dynamics
- Over time, as the electron moves closer to the positive terminal, it continues to experience similar forces—repulsion from negative and attraction towards positive—leading to ongoing movement.
- Each time it moves further along its path, both forces adjust based on its new position relative to each terminal.
- This continuous cycle results in gradual movement toward completing an electrical circuit by returning electrons back to fill "holes" left by other electrons moving away from terminals.
Understanding Electric Field Lines
- It’s crucial to grasp that it's these electric forces generated by both terminals that facilitate electron movement.
- When visualizing this process over time, we can represent it as "lines of force," illustrating how an electron's trajectory allows it to return effectively to complete a circuit.
Anticipating Future Concepts: Electric Fields
- Looking ahead, future discussions will focus on understanding how electric forces act on free electrons throughout space around a battery.
- This exploration will lead into concepts regarding all possible lines of force surrounding an electrical source—collectively known as an electric field.
- These lines represent various points where electric forces are present in space around charged objects.
Summary of Course Insights and Future Plans
Key Takeaways from the Course
- The course aimed to provide a foundational understanding of electric force, emphasizing its role in the movement of free electrons in electronic circuits. This concept is crucial for future lessons.
- The instructor expressed gratitude towards the growing audience, noting an unexpected level of interest and positive feedback on the courses, which has been encouraging.
- The instructor is passionate about creating these courses but highlighted that they require significant time investment—approximately 100 hours per course—which limits frequency.
- Due to time constraints, the instructor cannot produce weekly courses; however, they aim to release around ten episodes throughout 2017 despite being overwhelmed with commitments.
- The lack of free time is identified as a major challenge for content creation. The instructor plans to continue working on various projects, including fusion technology and equipment reviews.
Looking Ahead
- In closing, the instructor wished everyone an excellent year ahead and encouraged patience regarding upcoming content releases.