Muddiest Point- Phase Diagrams I: Eutectic Calculations and Lever Rule
Phase Diagrams: Calculations and Lever Rule
In this screencast, the speaker explains phase diagrams, their importance in understanding material systems, and how to interpret them. The speaker also defines key terms such as alpha, beta, eutectic reaction, and lever rule.
Introduction to Phase Diagrams
- A phase is a physically homogenous part of a material system.
- Phase diagrams tell us which phases are present at a given temperature and nominal composition.
- Most alloys have two or more phases because the properties improve.
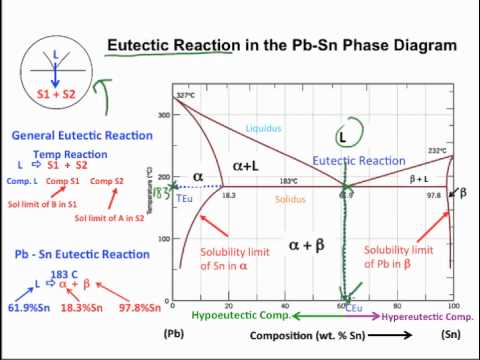
- A metal-metal phase diagram shows which phases are present at a given temperature on the y-axis and nominal composition on the x-axis.
Single Phase Regions
- The Alpha region is a single-phase region that is lead-rich.
- The Beta region is a single-phase region that is tin-rich.
- The liquid region is a single-phase region made of liquid components lead and tin.
Two-Phase Regions
- The Alpha + Liquid region is a two-phase slush consisting of solid alpha and liquid.
- The Beta + Liquid region is also a two-phase slush consisting of solid beta and liquid.
- The Alpha + Beta region consists of combinations of alpha and beta in solid form.
Eutectic Reaction
- By definition, the eutectic reaction occurs when liquid cools directly into the two-phase solid region of eutectic alpha plus beta without going through either the Alpha + Liquid or Beta + Liquid regions first.
- Eutectic temperature is the temperature at which the eutectic reaction takes place.
- To find it, start at this point (eutectic point) then head over to the y-axis which gives us temperature.
- This value right here (in our case 183 degrees Celsius) is our eutectic temperature.
- Eutectic composition is the nominal composition of the eutectic reaction.
- To find it, start again at this point and go straight down to the x-axis.
- The value where you land is your eutectic composition.
- The chemical composition of each phase at the eutectic composition is found by using the solubility limits.
Lever Rule
- The lever rule is used to determine the weight fraction of each phase in a two-phase region.
- It states that the ratio of distances from a point on a tie line to either end represents the weight fraction of each phase.
- This can be used to find chemical compositions and phase fractions from a phase diagram.
Conclusion
- Phase diagrams are important for understanding material systems and determining which phases are present at different temperatures and nominal compositions.
- Key terms such as alpha, beta, eutectic reaction, and lever rule were defined and explained.
Eutectic Composition
In this section, the speaker discusses the eutectic composition and its significance in phase diagrams.
Lead-Tin Eutectic Reaction
- The eutectic composition is given as 97.8% tin.
- The lead-tin eutectic reaction occurs at 183 degrees Celsius.
- The liquid phase cools down into the alpha plus beta region.
General and Specific Cases
- In general, the liquid cools into the two-phase solid region.
- In this specific case, lead is in solid two.
Hypo and Hyper Eutectic Compositions
- Any overall or nominal composition to the left of the eutectic composition is called hypo-eutectic.
- Any nominal composition to the right of the eutectic composition is called hyper-eutectic.
Example Phase Calculation
This section provides an example calculation for determining which phases are present, their chemical compositions, and their weight fractions.
Determining Phases Present
- At 40 weight percent tin and 60 weight percent lead, we find our point on the diagram by going down to 40% tin on x-axis and up to 150 degrees Celsius on y-axis.
- The intersection of these lines indicates that we are in the alpha plus beta region, so both phases are present.
Chemical Composition of Each Phase
- Alpha's composition can be found by going over from our point to solubility limit of tin in alpha at our temperature (150 degrees Celsius), which gives us a composition of 11% tin.
- Beta's composition can be found by going over to solubility limit of lead in beta at our temperature (150 degrees Celsius), which gives us a composition of 99% tin.
Phase Weight Fractions
- To find the weight fraction of alpha, we use the lever rule and calculate Q over P plus Q.
- To find the weight fraction of beta, we use the same formula but with P instead of Q.
- The weight fraction of alpha is 0.66 and the weight fraction of beta is 0.33.
Using a Table for Calculations
This section provides an alternative method for organizing phase calculation answers using a table.
Organizing Answers in a Table
- A table can be used to organize answers for phase calculations.
- The table includes columns for each phase's composition and weight fraction.
- The lever rule can still be used to calculate weight fractions.
Chemical Composition and Phase Fraction
In this section, the speaker discusses how to find the chemical composition and phase fraction of alpha and beta in a eutectic reaction.
Approximating Phase Fractions
- The phase fraction of alpha can be approximated by dividing the distance of composition beta from the eutectic point by the total distance of composition alpha.
- Using this formula, the phase fraction of alpha is found to be 0.67.
- Once we have our phase fraction of alpha, we can use another formula to find our phase fraction of beta. Plugging in all our numbers, we can see that our phase fraction of beta would be 0.33.
Organizing Values in a Table
- Organizing values in a table can make calculations easier and help approximate values more accurately.
Recap
- The speaker has discussed what beta and alpha represent, explained all about the eutectic reaction and its terminology, and demonstrated how to find chemical composition and phase fractions from it. All muddiest points have been addressed.