XPS Spectra for p, d, and f-orbitals
Understanding XPS Analysis: Peak Splitting and Intensity Ratios
Introduction to Orbital Splitting in XPS
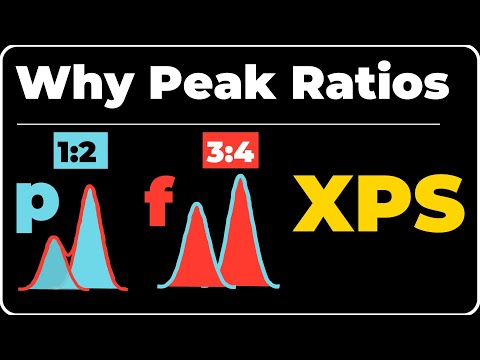
- The discussion begins with the explanation of why p, d, and f orbitals split into multiple peaks during X-ray Photoelectron Spectroscopy (XPS) analysis. It highlights the intensity ratios observed for these orbitals.
- The speaker introduces the concept of total angular momentum (J), which is a combination of orbital angular momentum and spin angular momentum, crucial for understanding peak splitting.
Angular Momentum and Its Role
- The relationship between orbital angular momentum (L) and its implications on peak intensities is discussed. For example, L = 0 results in a single peak for s orbitals.
- An overview of electron behavior around the nucleus is provided, emphasizing both orbital motion and spin as contributors to total angular momentum.
JJ Coupling Explained
- The concept of JJ coupling in quantum mechanics is introduced, explaining how it relates to spin-orbit splitting or doublets in spectral lines.
- For p orbitals with L = 1, the presence of non-zero angular momentum leads to observable doublets in their spectra.
Deriving Intensity Ratios
- A visual representation shows that smaller peaks indicate lower concentration elements; this observation supports the derived intensity ratios.
- The nomenclature used to represent XPS peaks (n l j notation) is explained further, clarifying how principal quantum numbers relate to observed spectral features.
Proving P Orbital Intensity Ratios
- To demonstrate why p orbitals have an intensity ratio of 1:2, a formula involving degeneracy (2J + 1) is referenced.
- Using oxygen as an example, the speaker illustrates how splitting occurs within p orbitals leading to specific intensity ratios based on electron configurations.
D Orbitals Intensity Ratio Calculation
- Similar calculations are applied to d orbitals where J values lead to a derived ratio of 6:4 or simplified down to 3:2 due to ten electrons present in d orbitals.