Episódio 16 - A Misteriosa Rotação (Momento Angular/Magnético de Spin)
Introduction
The video introduces the topic of spin in atomic systems, specifically focusing on subatomic particles and their intriguing properties.
Understanding Spin in Atomic Systems
- Spin is explained differently from conventional teachings, emphasizing a unique perspective based on real understanding.
- Historical background of spin is discussed, tracing back to the proposal by Goldsmith and Uhlenbeck in 1925 regarding electron behavior in complex atomic spectra.
- The proposal of spin stemmed from difficulties in explaining spectral lines in atoms beyond hydrogen using Einstein's theory, leading to the concept of intrinsic angular momentum.
Origin of Spin Concept
Delving into the origins and development of the spin concept within quantum mechanics.
Proposal by Goldsmith and Uhlenbeck
- Goldsmith and Uhlenbeck proposed that electrons possess both orbital angular momentum and intrinsic angular momentum (spin), revolutionizing atomic structure understanding.
- Introduction of electron's intrinsic angular momentum distinct from its orbital motion paved the way for explaining spectral line occurrences.
Evolution of Quantum Mechanics with Spin
Evolution of quantum mechanics incorporating spin as a fundamental property.
Significance of Electron Spin
- Electron's intrinsic angular momentum, known as spin, was attributed to characteristics like mass and charge, fundamentally altering atomic models.
- Analogies between electron spin and Earth's rotation highlight conceptual similarities without implying literal spinning motion.
Quantum Mechanics Advancements
Advancements in quantum mechanics integrating relativistic properties and introducing new quantum numbers related to electron behavior.
Contributions by Dirac and Others
- Dirac's modifications to wave mechanics introduced relativistic aspects, leading to predictions of electron spin through additional quantum numbers.
- Inclusion of electron spin as a fundamental property alongside other quantum restrictions reshaped atomic theory perception significantly.
Experimental Validation
Experimental validation preceding theoretical proposals regarding electron spin.
Experiment by Stern-Gerlach
- Stern-Gerlach experiment demonstrated silver atoms' magnetic properties aligning with electron angular momentum under a magnetic field, confirming the existence of electron spin.
New Section
In this section, the discussion revolves around the interpretation of magnetic moments in atoms, particularly focusing on silver atoms and their behavior under different conditions.
Interpreting Magnetic Moments in Atoms
- When observing a screen with silver atoms projected onto it, a clear separation between them was noted based on their magnetic moments.
- The distinct separation observed was attributed to the interpretation of orbital magnetic moments, leading to two extreme values being identified.
- By examining concentrated clusters of silver atoms at different positions, the impact of external factors like sulfur from smoking on the darkening of silver was highlighted.
- The interaction between sulfur and silver causing darkening led to initial misinterpretation as an orbital magnetic moment due to using single-electron silver atoms intentionally.
- Initial interpretations linked the observed phenomena to orbital angular momentum; however, advancements in quantum mechanics revealed that it was actually due to electron spin rather than orbital motion.
Exploring Spin Angular Momentum
This segment delves into the concept of spin angular momentum in quantum mechanics and its implications for understanding atomic properties.
Understanding Spin Angular Momentum
- Quantum mechanics reveals that electrons possess spin angular momentum independent of their orbital motion, contrary to earlier assumptions.
- The discovery that electron spin contributes significantly to magnetic properties highlights a shift in understanding from solely focusing on orbital angular momentum.
- Historical experiments by Stern and Gerlach inadvertently confirmed the existence of electron spin rather than solely attributing observations to other factors like atomic structure or orbitals.
Properties of Spin Angular Momentum
This part explores how spin angular momentum influences atomic properties and necessitates modifications in wave functions for accurate representation.
Impact on Atomic Properties
- Spin angular momentum plays a crucial role in determining how particles interact with magnetic fields, leading to observable separations based on spin orientations.
- Modifying wave functions by introducing internal variables like omega enables a more comprehensive depiction of particle spins within quantum mechanics.
Atomic Spin and Magnetic Moment
In this section, the focus is on understanding the spin of an electron to determine the vector value of the magnetic moment. The discussion delves into calculating the spin angular momentum and its significance in quantum mechanics.
Understanding Spin Angular Momentum
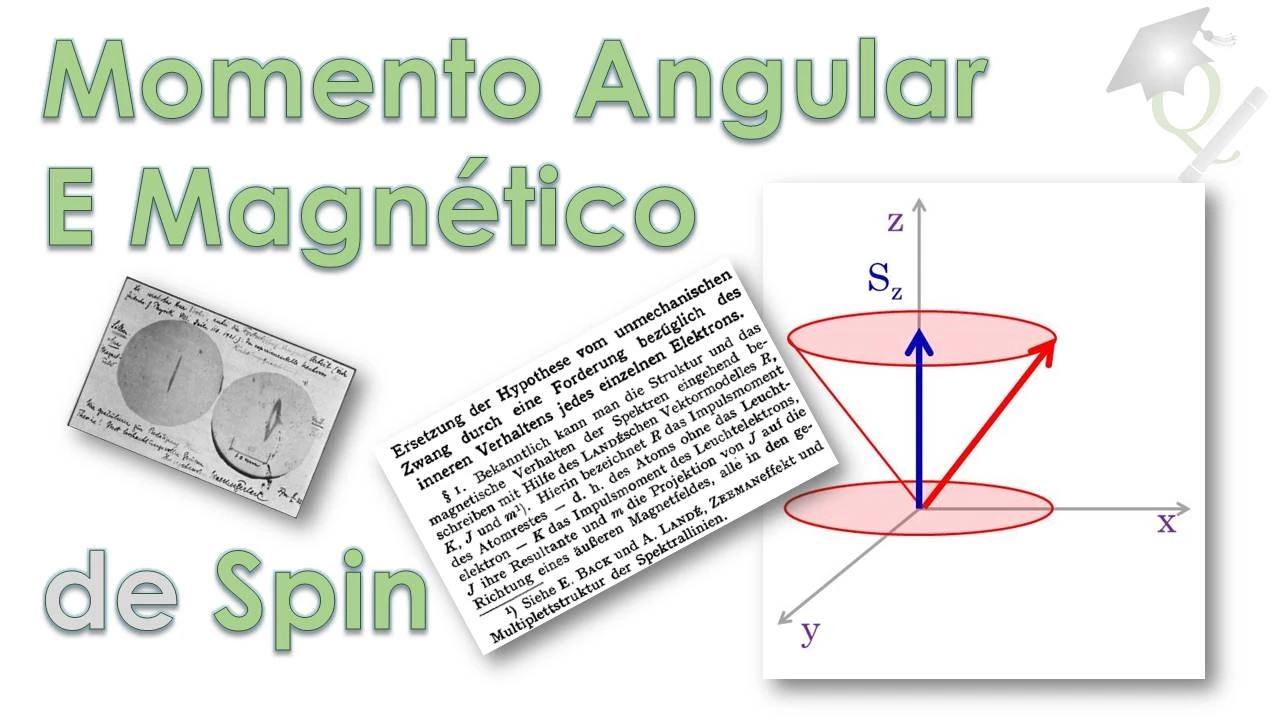
- The module of the spin vector is calculated as the square root of 0.75, representing a fundamental property in quantum mechanics.
- The spin vector can be visualized in a Cartesian plane, with projections on x, y, and z axes contributing to its definition.
- Projections of spin angular momentum on different axes are quantized, varying from -l to +l where l represents a quantum number.
Significance of Spin Projection
- The projection of spin angular momentum on the z-axis holds importance, indicating specific values such as -1/2 and +1/2.
- Understanding the concept of +/- 1/2 projections aids in interpreting the behavior and properties associated with electron spins.
Quantum Mechanics and Electron Spin
This segment explores how electron spins are constrained by quantum principles, emphasizing specific orientations and limitations within quantum mechanics.
Quantum Constraints on Electron Spin
- Electron spins cannot assume arbitrary values or orientations but are restricted to specific projections like +/- 1/2.
- Discussion clarifies misconceptions regarding electron spins arising from high school teachings about rotations and clockwise/counterclockwise concepts.
Unique Property of Electron Spin
- Electron spin is distinct from classical rotation, being an exclusive property governed by quantum mechanics rather than physical rotation.
- The intrinsic nature of electron spin remains enigmatic due to its unique properties not rooted in conventional rotational dynamics.
Measurement Challenges in Quantum Mechanics
Addressing measurement challenges related to determining spin projections highlights complexities inherent in quantum mechanics measurements.
Measurement Limitations
- Projections along different axes pose challenges as simultaneous measurements are constrained by uncertainty principles within quantum mechanics.
- Determining precise projections for electron spins around various axes reveals limitations in measuring multiple parameters simultaneously due to inherent uncertainties.
Momento Magnético e Spin
In this section, the discussion revolves around the resultant magnetic moment of spin in films, its association with angular momentum and magnetic moment, and its implications in experiments related to wars.
Momento Angular de Spin e Momento Magnético
- The formula for the magnetic moment is directly associated with the spin angular momentum.
- The projection of spin angular momentum on the z-axis yields a spin magnetic moment, indicated by a negative sign showing opposite directions.
Relação entre Momento Angular e Momento Magnético
- The relationship between spin angular momentum and spin magnetic moment is discussed, highlighting their opposing directions.
- Emphasis on the significance of the magnetic moment along the z-axis and its relation to specific values like half for an electron.
Momento Magnético de Spinning
This part delves into the concept of spinning magnetic moments for free electrons, emphasizing their negative balance indicating opposite directions as well as their connection to orbital angular momentum.
Conexão com o Momento Angular Orbital
- Discussion on how spinning magnetic moments relate to both spin and orbital angular moments.
- Explanation of how orbital magnetic moments depend on quantum numbers like ml, showcasing their significance in representing electrons.
Propriedades do Spin e Magnetismo
Here, the focus shifts towards interactions between different types of moments (spin vs. orbital), termed couplings, and clarifies misconceptions regarding electron spins being rotational.
Acoplamentos e Esclarecimentos
- Introduction to couplings between different types of moments like spin and orbital.
- Clarification that electron spins are intrinsic properties rather than rotational ones; emphasis on dispelling common misconceptions among students and educators.
Conclusão sobre o Spin Eletrônico
This final segment stresses the importance of understanding electron spins accurately to avoid perpetuating misconceptions within educational settings.
Importância da Correta Compreensão do Spin