9. Sinapsis Berardi 2022
Presentation on Neuronal Communication
In this section, the speaker delves into the intricacies of neuronal communication, focusing on the transmission of information between neurons and other cells.
Neuronal Networks and Synapses
- Neurons can establish 10,000 to 16,000 synaptic contacts each, leading to infinite communication networks.
- A synapse is a functional connection where a neuron contacts another specialized cell, such as a muscle or another neuron.
- Neurons can communicate through various types of synapses like axo-dendritic, axo-axonic, axo-somatic, or dendro-dendritic connections.
Classification of Synapses
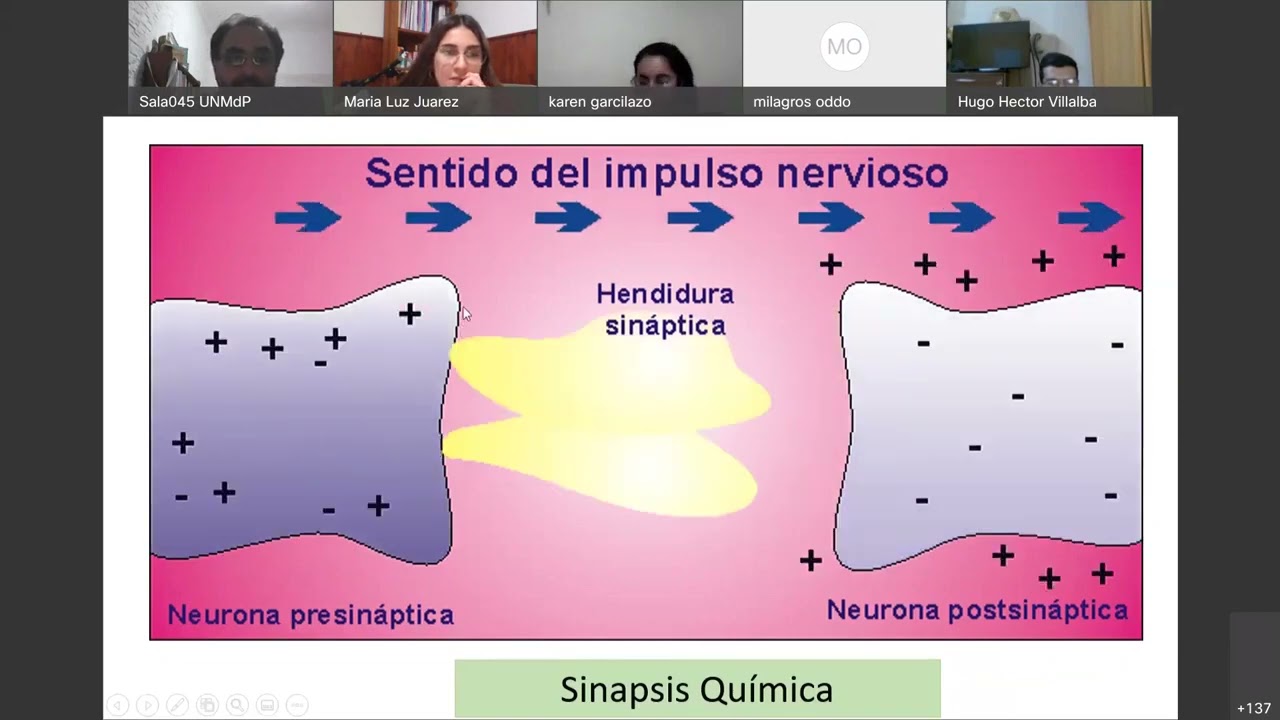
- Synapses are categorized into chemical, electrical, and mixed types. Chemical synapses predominate in the central nervous system and offer therapeutic intervention opportunities due to their complexity.
- Electrical synapses are simpler and faster but less modifiable therapeutically compared to chemical synapses.
Characteristics of Electrical Synapses
- Electrical synapses involve narrow gaps between presynaptic and postsynaptic neuron membranes with connexons facilitating ion passage for rapid communication.
Understanding Chemical Synapses
In this section, the discussion revolves around the process of chemical synapses and how they differ from electrical synapses in terms of structure and function.
Chemical Synapse Mechanism
- Inside the presynaptic neuron, there are round structures called synaptic vesicles containing neurotransmitters.
- The synaptic cleft between the presynaptic and postsynaptic membranes is over 20 nanometers wide, allowing neurotransmitters to diffuse.
- Upon arrival of an action potential at the axon terminal, voltage-gated calcium channels open, leading to calcium influx.
- Calcium entry triggers vesicle fusion with the presynaptic membrane, releasing neurotransmitters into the synaptic cleft for diffusion.
- Neurotransmitters do not cross the postsynaptic membrane but bind to receptors initiating intracellular changes.
Vesicle Fusion Process
- Calcium binding proteins facilitate vesicle fusion with both cytoplasmic and membrane proteins, enlarging the membrane upon fusion.
- Released neurotransmitters diffuse across the synaptic cleft to interact with specific receptors on the postsynaptic membrane.
Comparison: Electrical vs. Chemical Synapses
Contrasting electrical and chemical synapses highlights their distinct modes of signal transmission through direct connections or neurotransmitter release.
Signal Transmission Methods
- Electrical synapses rely on gap junctions for direct ion flow between neurons via connexons.
- In chemical synapses, neurotransmitters stored in synaptic vesicles are released upon vesicle-membrane fusion for inter-neuronal communication.
Functional Differences
- Neurotransmitter presence signifies a chemical synapse where neurotransmitter release is crucial for signal propagation.
Understanding Neurotransmitters in Synaptic Transmission
In this section, the discussion revolves around the role of neurotransmitters in synaptic transmission, focusing on their synthesis, storage, release, and interaction with receptors.
Neurotransmitter Functionality
- Neurotransmitters are chemical messengers called neurotransmitters that play a crucial role in synaptic communication.
- Neurotransmitters do not cross the postsynaptic membrane directly but are released from presynaptic neurons into the synaptic space to interact with receptors on the postsynaptic membrane.
- Most neurotransmitters do not enter the next neuron but instead bind to receptors on the postsynaptic membrane to initiate their action.
Vesicle Storage and Release
- Within axon terminals, numerous mitochondria and synaptic vesicles containing neurotransmitters are observed.
- The vesicles store neurotransmitters to prevent enzymatic degradation and ensure controlled release upon receiving an action potential.
Importance of Vesicular Storage
- Neurotransmitters are stored in vesicles to avoid enzymatic degradation within the cytoplasm and regulate their release in response to neuronal signaling.
- The process of packaging neurotransmitters into vesicles requires energy provided by mitochondria present at axon terminals.
Criteria for Neurotransmitter Identification
- The discovery of acetylcholine as a neurotransmitter highlighted specific criteria for identifying neurotransmitters: synthesis by neurons, storage in vesicles, release mechanisms, receptor interactions, and termination processes.
- These criteria form the mnemonic "El Salitre," representing synthesis (S), storage (A), release (L), interaction with receptors (I), and termination (T).
Classification of Neurotransmitters
Neurotransmitters and Receptors
In this section, the discussion revolves around neurotransmitters, distinguishing between true neurotransmitters like acetylcholine and putative neurotransmitters that do not meet all criteria but still exhibit actions.
Neurotransmitters Classification
- True neurotransmitters include acetylcholine, adrenaline, noradrenaline, glutamate, serotonin.
- Putative neurotransmitters are substances synthesized by neurons but may not be stored in vesicles; examples include nitric oxide and endocannabinoids.
- Nitric oxide is a gas synthesized but not storable; it travels quickly to penetrate another neuron. Endocannabinoids act on receptors similar to cannabis-derived components.
Neurotransmitter Storage and Release
This part delves into the storage of true neurotransmitters in synaptic vesicles to prevent enzymatic degradation and enable precise release upon nerve impulse arrival.
Vesicle Storage Mechanism
- True neurotransmitters are stored in synaptic vesicles to avoid enzymatic degradation and allow quantum release upon nerve impulse arrival.
- Fusion of synaptic vesicle with membranes requires two conditions: action potential reaching synaptic terminal for depolarization and opening of voltage-gated calcium channels facilitating vesicle function.
Neurotransmitter Receptors
The focus here is on classifying neurotransmitter receptors into ionotropic (ligand-gated ion channels) and metabotropic (G-protein-coupled receptors) categories based on their mechanisms of action.
Receptor Classification
- Neurotransmitter receptors are categorized as ionotropic (ligand-gated ion channels) or metabotropic (G-protein-coupled receptors).
- Ionotropic receptors directly form an ion channel upon activation by a ligand like the nicotinic receptor for acetylcholine.
Metabotropic Receptors
Metabotropic receptors differ from ionotropic ones as they activate intracellular proteins generating second messengers that propagate the signal within the neuron.
Metabotropic Receptor Function
- Metabotropic receptors trigger intracellular proteins to produce second messengers following neurotransmitter binding, extending the signaling cascade within neurons.
- Second messengers generated by metabotropic receptors amplify the initial signal transmitted by the neurotransmitter-receptor interaction.
Mensaje y Receptores Metabotrópicos
In this section, the discussion revolves around the interaction between neurotransmitters and metabotropic receptors, highlighting the process of message transmission within neurons.
Understanding Neurotransmitter-Receptor Interaction
- Metabotropic receptors are proteins that recognize neurotransmitters upon contact.
- Two scenarios are discussed: in metabotropic receptors, neurotransmitters do not enter but trigger a second messenger inside the receptor to continue the message.
- The synaptic cleft is emphasized as crucial in chemical synapses due to its role in separating pre- and post-synaptic membranes.
Segundo Mensajero y Receptores Metabotrópicos
This segment delves into second messengers and G proteins associated with metabotropic receptors, elucidating their roles in signal transduction pathways.
Significance of Second Messengers
- The protein G alongside metabotropic receptors activates a cascade of enzymes leading to the generation of second messengers like cAMP.
- Activation of protein G by neurotransmitters initiates cascades involving enzymes that produce secondary messengers such as cAMP and cyclic AMP.
Neurotransmisores Putativos y Potenciales de Acción
Here, putative neurotransmitters and action potentials are explored, shedding light on unique characteristics and functions within neural communication.
Insights on Neurotransmitters and Action Potentials
- Putative neurotransmitters lack specific criteria fulfillment; examples include lipophilic compounds like nitric oxide.
- Endocannabinoids like anandamide exemplify neurotransmitters synthesized and released upon electrical stimulation without vesicular storage.
Patologías Relacionadas con Canales de Calcio
This part discusses pathologies linked to voltage-gated calcium channels, emphasizing their impact on neuromuscular function.
Pathological Implications of Calcium Channels
- Autoantibodies targeting voltage-gated calcium channels can hinder neurotransmitter release, leading to muscle weakness as seen in Eaton-Lambert syndrome.
Neurotransmitters and Neuropeptides
In this section, the discussion revolves around the differences between small molecule neurotransmitters and large molecule neuropeptides in terms of synthesis, storage, release, and termination mechanisms.
Small Molecule Neurotransmitters vs. Neuropeptides
- Small molecule neurotransmitters are synthesized in the axon terminal close to the release site, while neuropeptides, being larger proteins, are synthesized in the neuronal soma where rough endoplasmic reticulum is present.
- Synthesized small molecule neurotransmitters are immediately stored in synaptic vesicles at the axon terminal for protection. In contrast, neuropeptides are also stored in synaptic vesicles after synthesis but require transportation from the soma to the axon terminal before release.
- Both types of neurotransmitters are released upon action potential arrival at the vesicle-containing zone where calcium channels open. Each neurotransmitter interacts with specific receptors on target cells post-synaptically.
- Small molecule neurotransmitters have two main termination mechanisms: reuptake by releasing neuron or uptake by nearby glial cells; whereas neuropeptides undergo enzymatic degradation post-receptor interaction to prevent continuous receptor activation.
Understanding Neurotransmitters and Synaptic Transmission
In this section, the speaker delves into the role of proteins in synaptic vesicles and axon terminals, highlighting the significance of calcium in protein interactions.
Proteins in Synaptic Vesicles and Axon Terminals
- Proteins are abundant in both the cytoplasm of synaptic vesicles and the membrane of axon terminals.
- Calcium facilitates protein interaction, including fusion proteins known for anchoring and fusion.
- Bacterial toxins like tetanus or botulism target these proteins, disrupting neurotransmitter release and causing paralysis.
Neurotransmitter Termination Mechanisms
The discussion shifts to neurotransmitter termination mechanisms, focusing on small molecule neurotransmitters' reuptake and degradation processes.
Small Molecule Neurotransmitters Termination
- Small molecule neurotransmitters like adrenaline or glutamate undergo reuptake by neurons or enzymatic degradation in the synaptic space.
- Two termination mechanisms exist: reuptake by neurons via transporters or enzymatic degradation within the synaptic space.
Differences in Neuropetides Termination
Contrasting small molecules, neuropetides diffuse from the synaptic space due to their size, emphasizing distinct termination mechanisms.
Neuropetides Termination
- Neuropetides lack reuptake transporters; hence they diffuse from the synaptic space for degradation.
Acetylcholine as a Neurotransmitter
Exploring acetylcholine as a neurotransmitter, detailing its synthesis, storage, release, receptor interaction, and termination mechanism.
Acetylcholine Functionality
- Acetylcholine is synthesized at nerve terminals through choline acetyltransferase enzyme action.
- Once synthesized, acetylcholine is stored in synaptic vesicles to prevent degradation outside.
Acetylcholinesterase Role in Acetylcholine Degradation
Highlighting acetylcholinesterase's crucial role in degrading acetylcholine post-receptor interaction for proper neuromuscular function.
Acetylcholinesterase Function
- Acetylcholinesterase degrades released acetylcholine into acetate and choline for recycling.
Understanding Myasthenia Gravis and Neurotransmitters
In this section, the discussion revolves around myasthenia gravis, a neuromuscular disease where antibodies target the muscle receptor for acetylcholine. The treatment involves inhibiting acetylcholinesterase to allow acetylcholine to compete with antibodies and promote muscle contraction.
Myasthenia Gravis and Acetylcholine Receptors
- Myasthenia gravis is a neuromuscular disease where antibodies target the nicotinic acetylcholine receptor in muscles.
- Treatment involves using an acetylcholinesterase inhibitor to prevent acetylcholine degradation, allowing it to compete with antibodies and facilitate muscle contraction.
Understanding Cholinergic Receptors
This section delves into cholinergic receptors, distinguishing between nicotinic and muscarinic receptors based on their stimulation sources and locations in the body.
Cholinergic Receptor Types
- Cholinergic receptors can be nicotinic (stimulated by nicotine) or muscarinic (stimulated by a fungal product called muscarine).
- Nicotinic receptors are ionotropic channels found at neuromuscular junctions in skeletal muscles, facilitating muscle contractions through sodium influx.
Distinguishing Nicotinic and Muscarinic Receptors
This segment highlights the differences between nicotinic and muscarinic receptors in terms of location, function, and involvement in voluntary versus involuntary muscle control.
Nicotinic vs. Muscarinic Receptors
- Nicotinic receptors are present in voluntary skeletal muscles for movement control, while muscarinic receptors are found in involuntary smooth muscles regulated by the parasympathetic system.
Role of Acetylcholine in Autonomic Nervous System
Exploring how acetylcholine functions within the autonomic nervous system, particularly its impact on different organs mediated by nicotinic or muscarinic receptors.
Acetylcholine Functionality
- In the autonomic nervous system, nicotinic receptors are located in voluntary skeletal muscles as well as sympathetic ganglia.
New Section
In this section, the discussion revolves around neurotransmitters such as glutamate and GABA, detailing their synthesis, storage, release mechanisms, and receptor types.
Glutamate as a Neurotransmitter
- Glutamate is derived from amino acids like glutamine or glucose. It is synthesized by neurons through an enzyme called glutaminase.
- Neurons follow a mnemonic rule of synthesis, storage in synaptic vesicles, and release via exocytosis upon calcium influx for glutamate.
- Glutamate can be recaptured by neurons for reuse or degraded within the neuron. Alternatively, glial cells transform recaptured glutamate into glutamine to pass back to neurons for further synthesis.
Receptors and Functions of Glutamate
- Glutamate acts on six different receptors: three ionotropic (AMPA, NMDA, kainate) and three metabotropic receptors. These receptors play crucial roles in memory formation and movement control.
- Metabotropic receptors generate second messengers upon activation by glutamate, influencing other neurotransmitters like GABA.
New Section
This segment delves into GABA as a neurotransmitter, its synthesis from glutamate via specific enzymes, storage in synaptic vesicles, release mechanisms through neuronal recapture or degradation.
GABA Synthesis and Function
- GABA is synthesized by neurons from glutamate using the enzyme glutamic acid decarboxylase. Once produced, it is stored in synaptic vesicles awaiting release triggered by calcium influx during action potentials.
- Upon release, GABA interacts with postsynaptic receptors; its termination mechanism involves neuronal recapture similar to that of glutamate.
GABA Receptors and Effects
- GABA has three receptor types; notably the ionotropic receptor Gabar which allows chloride ions to enter upon activation leading to membrane hyperpolarization due to negative charge influx.
- Benzodiazepines like Clonazepam act as agonists on Gabar receptors causing hyperpolarization effects similar to alcohol but with potential motor coordination issues if overused.
New Section
In this section, the discussion revolves around neurotransmitters in the central nervous system, focusing on glutamate and GABA as excitatory and inhibitory neurotransmitters, respectively.
Glutamate and GABA Neurotransmitters
- Glutamate is primarily depolarizing and excitatory in the central nervous system. However, its role as excitatory is not absolute.
- GABA is often referred to as the quintessential inhibitory neurotransmitter in the central nervous system, with its inhibitory nature determined by its receptor.
- Both glutamate and GABA are small-molecule neurotransmitters synthesized at nerve terminals.
New Section
This part delves into the termination mechanisms of acetylcholine, glutamate, and GABA, emphasizing their significance in neurobiology.
Termination Mechanisms of Neurotransmitters
- Acetylcholine's termination mechanism involves enzymatic degradation rather than generation.
- The enzyme responsible for degrading acetylcholine is crucial due to its implications for treatments lacking drugs that act on it.
- Understanding the termination mechanisms of glutamate and GABA involves neuronal adaptation or reuptake processes within neurons.
New Section
This segment transitions to discussing the synthesis of biogenic amines like dopamine, adrenaline, noradrenaline, serotonin, and histamine.
Synthesis of Biogenic Amines
- Biogenic amines such as dopamine play vital roles in mood regulation within the central nervous system.
- These neurotransmitters are synthesized at axon terminals alongside other small-molecule neurotransmitters like catecholamines.
- Key biogenic amines include dopamine, noradrenaline (epinephrine), serotonin (5-hydroxytryptamine), and histamine due to their amino group characteristics.
- Dopamine synthesis process initiates with phenylalanine conversion to tyrosine via phenylalanine hydroxylase enzyme activity.
New Section
In this section, the discussion revolves around the release of dopamine by specific neurons in the brain and its implications for conditions like Parkinson's disease.
Neurons Releasing Dopamine
- Neurons in the substantia nigra of the mesencephalon release dopamine towards the corpus striatum.
Treatment for Parkinson's Disease
- In Parkinson's disease, where there is a deficit of dopamine due to neuron death in the substantia nigra, treatment involves providing a precursor to dopamine.
- Levodopa serves as an immediate precursor to dopamine.
- The administration of levodopa allows living neurons to convert it into dopamine, replenishing its levels in the brain.
Catecholamines and Tyrosine
- Catecholamines derive from tyrosine rather than phenylalanine.
- Tyrosine acts as an intermediate step in catecholamine synthesis.
New Section
This segment delves into how understanding chemical synapses, particularly involving dopamine, aids in comprehending various treatments beyond Parkinson's disease.
Significance of Chemical Synapses
- Understanding dopaminergic synapses not only aids in treating Parkinson's but also provides insights into other treatments utilizing similar principles.
- Dopamine deficiency can be addressed by administering precursors or agonists that act on dopamine receptors.
- Chemical synapses involve multiple steps that can be targeted for intervention in various treatments.
New Section
The focus here is on the receptors involved in dopaminergic pathways and the mechanisms governing dopamine termination within neurons.
Dopamine Receptors and Termination Mechanisms
- Dopamine receptors are diverse with different subtypes regulating various functions within neural circuits.
- Following neurotransmission, neuronal reuptake terminates dopamine action through processes involving monoamine oxidase and catechol-O-methyltransferase enzymes.
New Section
Exploring how neurons releasing noradrenaline differ from those releasing dopamine, emphasizing enzymatic transformations crucial for neurotransmitter synthesis.
Noradrenergic Neurons
- Neurons releasing noradrenaline possess dopamine beta-hydroxylase enzyme converting all available dopamine into noradrenaline before release.
- Such neurons are termed noradrenergic due to their predominant release of noradrenaline over dopamine.
Understanding Neurotransmitters and Receptors
In this section, the discussion revolves around neurotransmitters such as adrenaline and noradrenaline, their reuptake inhibition by cocaine, and the impact on synaptic activity.
Neurotransmitter Actions
- Cocaine inhibits the reuptake of adrenaline and noradrenaline, leading to increased activity due to more neurotransmitters in the synapse.
- Adrenaline and noradrenaline act on sympathetic receptors (Alpha and Beta), causing effects like tachycardia and vasoconstriction.
- Both Alpha and Beta receptors are metabotropic, acting through second messengers. An example of Alpha 1 receptor location is in arterioles for vasoconstriction.
Receptor Specificity
- Adrenaline acts on Beta 1 receptors in the heart, generating second messengers internally. An example of Beta 2 receptor activation is bronchodilation in response to Salbutamol.
- The termination mechanism for neurotransmitters involves reuptake by neurons or degradation by enzymes like MAO and COMT.
Serotonin Function
- Serotonin is a neurotransmitter affecting mood regulation, synthesized from tryptophan. It is stored in synaptic vesicles, released, acts on receptors, then recaptured by neurons.
- Antidepressants inhibit serotonin reuptake to increase its availability in synapses for improved mood regulation.
Neuropetides vs. Neurotransmitters
This part delves into the differences between small molecule neurotransmitters like dopamine and larger molecules known as neuropeptides regarding synthesis, storage, release mechanisms, and termination processes.
Neuropeptide Characteristics
- Neuropeptides are synthesized as prepropeptides in neuronal soma before storage in vesicles where they mature during transport to terminals.
- Upon release, neuropeptides interact with receptors but do not undergo reuptake; instead, they degrade or diffuse from the synaptic space for subsequent breakdown.
Neurotransmitter Comparison