Problema 1: Solución al diseño de una columna Absorción. Parte I
Design of Absorption Column
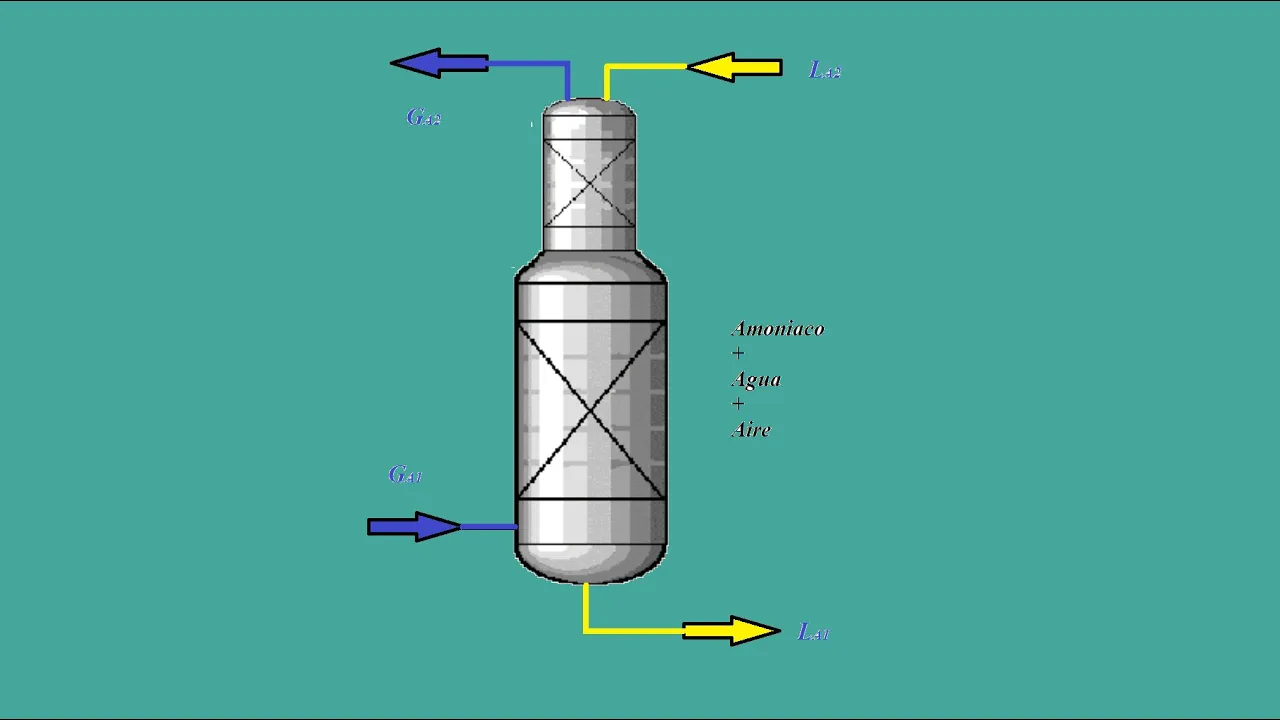
In this section, the speaker introduces a problem related to designing an absorption column packed with ceramic rings. The design parameters and requirements for separating ammonia from an air stream are discussed.
Design Parameters and Requirements
- The column needs to be packed with 5mm diameter ceramic rings with a characterization factor of 2025.
- A pressure drop of 200 pascals per meter is required to separate 99% of ammonia from an air stream.
- Liquid (water) enters the column at a rate of 650 liters per hour, in countercurrent flow, free of ammonia.
- Operating conditions include a separation efficiency of 99%, operating at 20 degrees Celsius and one atmosphere pressure.
Data Analysis and Calculations
This section delves into data analysis, including equilibrium data, transfer coefficients, and resistance calculations necessary for the design process.
Data Analysis
- Equilibrium data is presented in a table format for the given conditions.
- The volumetric average gas phase transfer coefficient is determined as 165 kmol/(h·m³·atm).
- Viscosity of the liquid phase remains constant at 0.0012 kg/(m·s), while liquid density is estimated at 0.98 g/cm³.
Component Analysis and Calculations
This part focuses on component analysis involving molecular weights, determining recovered ammonia mass per unit water fed, and calculating gas phase flow rate.
Component Analysis
- Molecular weights are provided: ammonia (17), inert gas in the gas phase (29 - air), inert liquid phase (18 - water).
- Calculation involves determining recovered ammonia mass per unit water fed and the exit gas phase flow rate.
Molar Relationships and Final Calculations
This segment covers molar relationships between components, deriving key values such as mole fractions for further calculations.
Molar Relationships
- Derivation of mole fraction relationships aids in calculating values like recovered ammonia mass per unit water fed.
Problems and Calculations in Chemical Engineering
In this section, the speaker discusses calculations related to chemical engineering problems, focusing on the composition of ammonia and liquid density.
Understanding L2 Calculation
- The speaker explains that L2 can be deduced as ls multiplied by one plus x a 2, where x a 2 is equal to 0, resulting in L2 being equal to ls.
Calculation of L2
- L2 is described as ls divided by liquid molar volume, simplifying to ls divided by liquid density since L2 equals ls.
Unit Conversion for Density Calculation
- Converting volume from liters per hour to cubic meters ensures homogeneous units. It is noted that 1 cubic meter equals 1000 liters.
Incorporating Molecular Weight for Density Calculation
- To determine the molar density of liquid, molecular weight plays a crucial role. The molecular weight is calculated as 18 kilograms per kilomole.
Balancing Material in Absorption Columns
This part delves into material balance within absorption columns, emphasizing the importance of composition balance.
Material Balance Fundamentals
- The material balance equation for an absorption column involves inert gas flow rate and mol ratio gradients in gas and liquid phases.
Composition Balance Equations
- Balancing compositions entails comparing maximum and minimum values. By setting x a 2 to zero, x a 1 can be determined using relevant equations.
Deriving Composition Ratios
- Through manipulation of equations, x a 1 can be expressed in terms of maximum and minimum values. Simplification leads to obtaining x a 1 based on given parameters.
Graphing Phase Liquid Composition
Graphical representation aids in visualizing phase liquid composition relationships.
Graphing Pressure Relationships
- Pressure relationships are graphed based on partial pressures of components A and B. Utilizing Dalton's law allows for determining total pressure and individual component pressures.
Pressure Conversion Calculations
- Converting atmospheric pressure to millimeters of mercury facilitates pressure calculations. Substituting known values yields accurate results for further analysis.
Determining Maximum Liquid Phase Composition
Determining maximum liquid phase composition involves graphical analysis.
Graphical Analysis Process
- Plotting equilibrium lines aids in identifying constant values like y a 1 max. Intersecting these lines helps find corresponding maximum compositions accurately.
Calculating Maximum Composition Value
- By locating intersection points on the graph, specific values such as x a 1 max can be determined precisely through graphical methods.
Calculating Relationship Values
Calculating relationship values involves applying derived formulas accurately.
Applying Formulas
- Utilizing established formulas enables precise calculation of relationship values like x a 1 based on given data points.
New Section
In this section, the speaker discusses the value of X and its relation to kilomoles.
Value of X and Kilomoles
- The value of X is 1, which equals 0.6526 kilomoles per kilomol dls.
- Converting kilomoles to kilograms: 0.6526 kilomoles/kilomol dls equals 17.7 kilograms/kilomol.
- Conversion to meters cubed: Consider molecular weight and liquid density for conversion.
New Section
This part delves into calculations involving water's molecular weight and density.
Water Molecular Weight and Density Calculations
- Calculation involving water's molecular weight (18) and density in kg/m^3.
- Further calculations: 980 kg/m^3 divided by cubic meters yields specific units.
New Section
The discussion shifts towards volumetric calculations involving water.
Volumetric Calculations for Water
- Multiplying values to obtain 6.04046 kg/m^3 for water.
- Finalizing volumetric flow rate calculation for G2 per hour based on previous steps.
New Section
Exploring the relationship between GS, G2, and densities in gas phase calculations.
Gas Phase Densities Calculation
- Deriving GS from known equations involving x values.
- Expressing G2 in terms of gas densities for further calculations.
New Section
Delving into molar relationships and simplifications in the context of ammonia-free columns.
Molar Relationships Simplification
- Utilizing known values like L2 = LS to simplify equations.
- Applying numerical values leads to a calculated hourly flow rate of 18.81 mol/hour.
New Section
Determining the volumetric flow rate by calculating gas total density at exit conditions.
Volumetric Flow Rate Calculation
- Using PT/RT formula with given pressure and temperature conditions.
Detailed Analysis of Column Diameter Calculation
In this section, the speaker discusses the calculation of the column diameter based on the volumetric flow rate of the gas phase leaving the column.
Calculating Column Diameter
- The cross-sectional area at the bottom or top of the column controls exceptions.
- The cross-sectional area remains constant whether at the bottom or top. The column diameter can be determined by a mass flow ratio relationship.
- For absorption processes, significant transfer occurs at the bottom. Calculations involve G1 and G1 prime ratios due to absorption nature.
- Determining G1 involves multiplying G1 by its molecular weight. Molecular weight calculations are crucial for accurate results.
- Molecular weights for gases like A and B (ammonia in this case) are essential for precise calculations.
Calculation of Gas Phase Load and Prime State Load
This part focuses on calculating the gas phase load and prime state load to determine the cross-sectional area within the column.
Gas Phase Load Calculation
- Gas 1 calculation involves multiplying GS by specific values to obtain accurate results.
- Detailed calculation steps lead to determining 21.33 kilomoles of G1 per hour.
- Final result shows 584.96 kilograms of G1 per hour, a crucial value for further calculations.
Prime State Load Determination
- Finding G1 prime state load is vital for establishing the cross-sectional area within the column.