Fattyacid catabolism Part 1
Understanding Fatty Acid Catabolism
Overview of Learning Goals
- The chapter focuses on fatty acid catabolism, detailing how fats are digested in animals. Understanding the mobilization and transport of fats in tissues is essential for this topic.
- By the end of the chapter, learners will grasp the concept of ketone bodies, their production, and significance. The chapter is divided into three main sections: digestion, mobilization, and transport of fats.
Digestion and Mobilization of Fats
- The first section discusses the sources of fatty acids and their routes to oxidation sites. Fatty acid oxidation occurs in three stages: beta oxidation converts long-chain fatty acids to acetyl-CoA.
- Acetyl-CoA undergoes further oxidation in the citric acid cycle, generating electrons that enter the electron transport chain within mitochondria. This process is briefly covered alongside beta oxidation specifics.
Energy Source from Fats
- Oxidation of fatty acids serves as a major energy source; approximately one-third of energy needs come from dietary triacylglycerols (TAG). TAG consists of glycerol bonded with three fatty acids, providing significant energy for organs like the liver and heart.
- Hibernating animals rely heavily on fat as an energy source due to its high caloric content compared to carbohydrates or proteins. Adipocytes store large amounts of TAG as fat droplets that can fill cells almost entirely.
Advantages of Fat Storage
- Fats offer efficient fuel storage advantages over polysaccharides due to higher energy per carbon atom and reduced solvation effects since they are non-polar molecules. This makes them more suitable for long-term energy storage compared to carbohydrates which are more hydrated and less concentrated in terms of stored energy.
- Triacylglycerols have a high caloric density because their long hydrocarbon chains yield more than twice the energy upon complete oxidation compared to equal weights of carbohydrates or proteins. Their insolubility also prevents osmotic pressure issues within cells during storage processes.
Mobilization Process
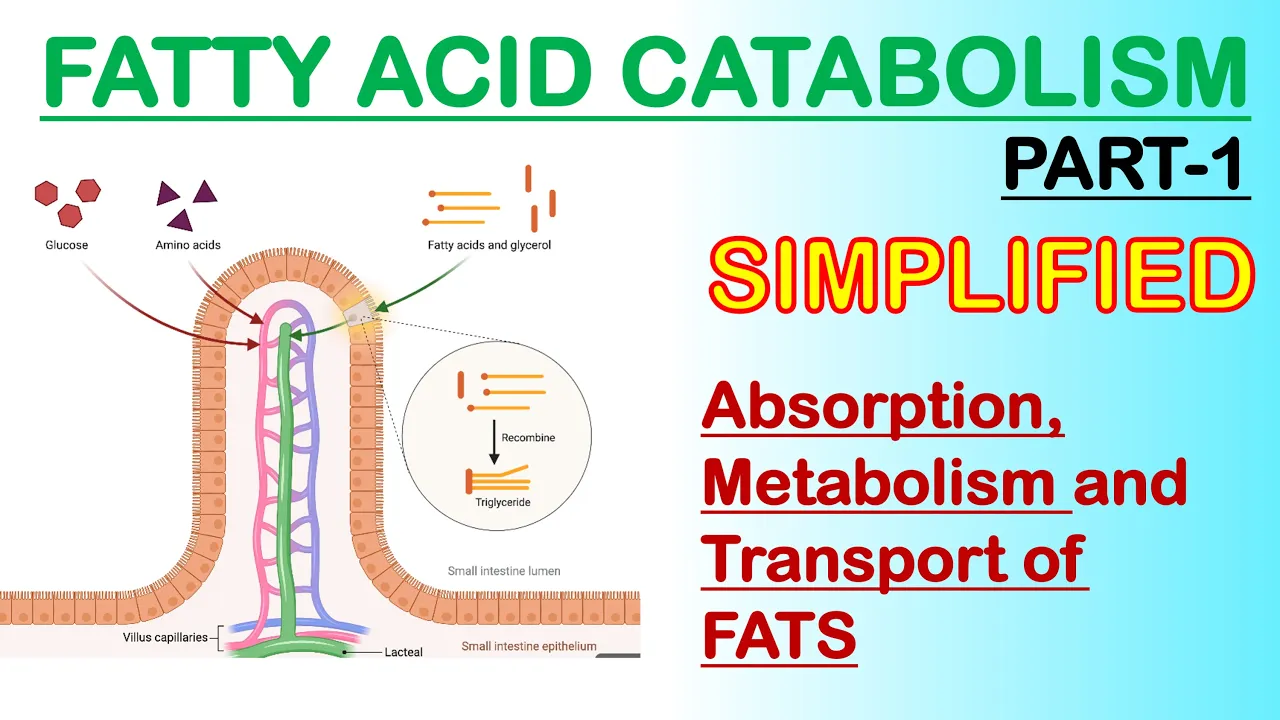
- Dietary fats must be converted into micelles before absorption through intestinal walls; bile salts facilitate this process by acting as biological detergents that emulsify fats into smaller particles accessible for lipase action. This conversion significantly enhances lipid accessibility for digestion by water-soluble enzymes like lipases.
Understanding Lipoproteins and Fat Mobilization
The Role of Apolipoproteins in Lipoprotein Formation
- Apolipoproteins are proteins that, when free of lipids, are termed apolipoproteins; upon binding with lipids, they form lipoproteins.
- Chylomicrons, which consist of triacylglycerols and apolipoproteins (like ApoC2), transport fats from the intestine into the bloodstream for distribution to muscle and adipose tissues.
- These chylomicrons travel through the bloodstream primarily to adipose tissues where fats are stored.
Fat Utilization in Different Tissues
- In muscle tissues, fatty acids from chylomicrons are oxidized for energy, while in adipose tissues, they undergo re-esterification for storage.
- After delivering their contents, remnants of chylomicrons (depleted of glycerols but containing cholesterol and apolipoproteins) return to the liver via blood circulation.
Energy Production from Triacylglycerols
- The liver processes these remnants through endocytosis; triacylglycerols can be oxidized for energy or serve as precursors for ketone body synthesis.
- Chylomicrons comprise about 80% triacylglycerols and cholesterol esters, with phospholipids forming a surface layer that interacts with aqueous environments.
Hormonal Regulation of Fat Mobilization
- Hormones trigger the mobilization of stored triacylglycerols; low glucose levels stimulate glucagon release which activates G protein-coupled receptors (GPCR).
- Activation of GPCR leads to adenylyl cyclase activation and increased cyclic AMP (cAMP), subsequently activating Protein Kinase A (PKA).
Mechanism Behind Lipid Mobilization
- PKA phosphorylates hormone-sensitive lipase (HSL) and perilipin; perilipin's phosphorylation allows access to lipid droplets by dissociating from CGI-58.
- Once activated by CGI-58, Adipose Triglyceride Lipase (ATGL) converts triacylglycerols into diacylglycerols releasing one fatty acid.
Breakdown Process of Stored Fats
- HSL further hydrolyzes diacylglycerols into monoacylglycerols releasing another fatty acid.
- Monoacyl glycerol lipase (MGL) then converts monoacyl glycerol into free fatty acids and glycerol.
Transporting Fatty Acids to Muscle Cells
- Resulting free fatty acids exit adipocytes into the bloodstream bound to serum albumin which transports them to myocytes (muscle cells).
- Inside myocytes, fatty acids enter through specific transporter proteins where they undergo beta oxidation for ATP production during energy crises.
Key Players in Fat Transport and Mobilization
- Apolipoproteins play a crucial role in transporting various lipids between organs when combined with lipids forming different classes of lipoproteins.
- Lipoprotein lipase is essential as it hydrolyzes triacylglycerols into usable forms: fatty acids and glycerol.
Understanding Lipid Metabolism
Lipolysis and Glycerol Conversion
- Hormone-sensitive lipase converts triglycerides into diglycerides, while monoacylglycerol lipase further breaks them down to free fatty acids and glycerol.
- Approximately 95% of the energy from triglycerides is derived from long-chain fatty acids, with only 5% coming from glycerol.
- Glycerol released by lipases is phosphorylated by glycerol kinase to form glycerol 3-phosphate, which then enters glycolysis through subsequent enzymatic reactions.
Fatty Acid Activation and Transport
- Once fatty acids enter myocytes, they are activated for transport into mitochondria where beta oxidation occurs.
- Fatty acids with chains shorter than 12 carbons can passively diffuse into mitochondria; those longer require a carnitine shuttle for transport.
- The activation of fatty acids involves conversion to fatty acyl-CoA via the enzyme fatty acyl-CoA synthetase, utilizing ATP in the process.
Energetics of Fatty Acid Activation
- The conversion of fatty acid to adenylate releases pyrophosphate, which is hydrolyzed to two phosphate molecules, contributing significantly to the overall energetics (total ΔG = -34 kJ/mol).
- This step emphasizes that fats are degraded into free fatty acids and glycerol in adipocytes before being transported for energy production.
Beta Oxidation Process
- Beta oxidation occurs in mitochondria; smaller fatty acids diffuse freely while larger ones utilize acyl-carnitine transporters.
- Acyl-carnitine formation allows larger fatty acids to be carried across mitochondrial membranes for oxidation.
Role of Carnitine in Fatty Acid Transport
- Fatty acyl-CoA formed in the cytosol attaches transiently to carnitine's hydroxyl group for mitochondrial entry.
Carnitine Shuttle and Fatty Acid Oxidation Overview
The Carnitine Shuttle Process
- The final step of the carnitine shuttle involves transferring the fatty acyl group from carnitine to intra-mitochondrial coenzyme A (CoA), resulting in the formation of fatty acyl-CoA in the matrix.
- The process begins with converting fatty acyl-CoA into acyl-carnitine, allowing it to be transported across the intermembrane space into the mitochondrial matrix, where it is converted back into acyl-CoA.
Stages of Fatty Acid Oxidation
Stage One: Beta-Oxidation
- Fatty acid oxidation occurs in three stages; stage one involves oxidative conversion of two-carbon units into acetyl-CoA through beta-oxidation, generating NADH and FADH2.
- In this stage, fatty acids undergo oxidative removal of successive two-carbon units as acetyl-CoA. For example, a 16-carbon palmitoyl-CoA undergoes seven cycles to yield eight acetyl-CoA molecules.
Stage Two: Citric Acid Cycle
- Acetyl groups from acetyl-CoA are oxidized to carbon dioxide via the citric acid cycle, producing additional NADH and FADH2. Each cycle generates two carbon dioxide molecules.
- From eight acetyl-CoAs entering the citric acid cycle, a total of 16 carbon dioxide molecules and 64 electrons are produced.
Stage Three: Electron Transport Chain
- The reduced electron carriers (NADH and FADH2) generated from stages one and two donate electrons to the mitochondrial respiratory chain (electron transfer chain), ultimately leading to ATP production through ADP phosphorylation.
Detailed Mechanism of Beta-Oxidation
Initial Steps with Palmitoyl CoA
- Upon entering the mitochondrial matrix, palmitoyl CoA is acted upon by acyl CoA dehydrogenase which oxidizes it to trans-delta 2-enoyl CoA using FAD as a cofactor.
- This reaction introduces a double bond between alpha and beta carbons, forming trans-delta 2-enoyl CoA.
Subsequent Reactions
- Enol CoA hydratase adds water across the double bond in trans-delta 2-enoyl CoA to form L-beta-hydroxyacyl CoA.
- L-beta-hydroxyacyl CoA is then oxidized by beta-hydroxyacyl CoA dehydrogenase to produce beta-ketoacyl CoA while reducing NAD+ to NADH.
Final Step