Chapter 9 Part 4
Welcome and Introduction to Citric Acid Cycle
In this section, the speaker introduces the citric acid cycle and highlights the generation of high-energy electron carriers through enzymatic reactions.
Key Concepts of Citric Acid Cycle
- The citric acid cycle involves eight enzymatic reactions that produce high-energy electron carriers like NADH and FADH2.
- Energy from acetyl-CoA is extracted through a series of reactions, leading to the formation of NADH and FADH2.
- While ATP is not directly produced in the citric acid cycle, GTP can be generated, which can later contribute to ATP production.
- Both ATP and GTP store energy in their phosphate groups, making them essential for powering energetically unfavorable reactions in cells.
- The citric acid cycle generates crucial molecules that are utilized in oxidative phosphorylation during cellular respiration.
Energy Generation and Carbon Transformation in Citric Acid Cycle
This part delves into how carbon atoms from pyruvate are transformed into acetyl groups, emphasizing the role of key molecules like NADH and FADH2.
Carbon Transformation Process
- Pyruvate undergoes oxidation to form acetyl-CoA, losing a carbon atom as CO2 waste expelled by breathing.
- Acetyl group combines with oxaloacetate to form citrate, initiating a series of reactions that lead to energy extraction.
- Each turn of the citric acid cycle results in the loss of carbons from acetyl groups and the generation of NADH, FADH2, and ATP or GTP equivalents.
- When starting with glucose instead of pyruvate, all quantities in the cycle are doubled due to two pyruvate molecules resulting from glycolysis breakdown.
- The citric acid cycle enables the capture of residual energy present in glucose molecules through multiple turns involving various energy carriers.
Role of High-Energy Electron Carriers in Cellular Respiration
This segment focuses on elucidating how high-energy electron carriers like NADH and FADH2 play a pivotal role in transferring extracted energy for further utilization.
Significance of Electron Carriers
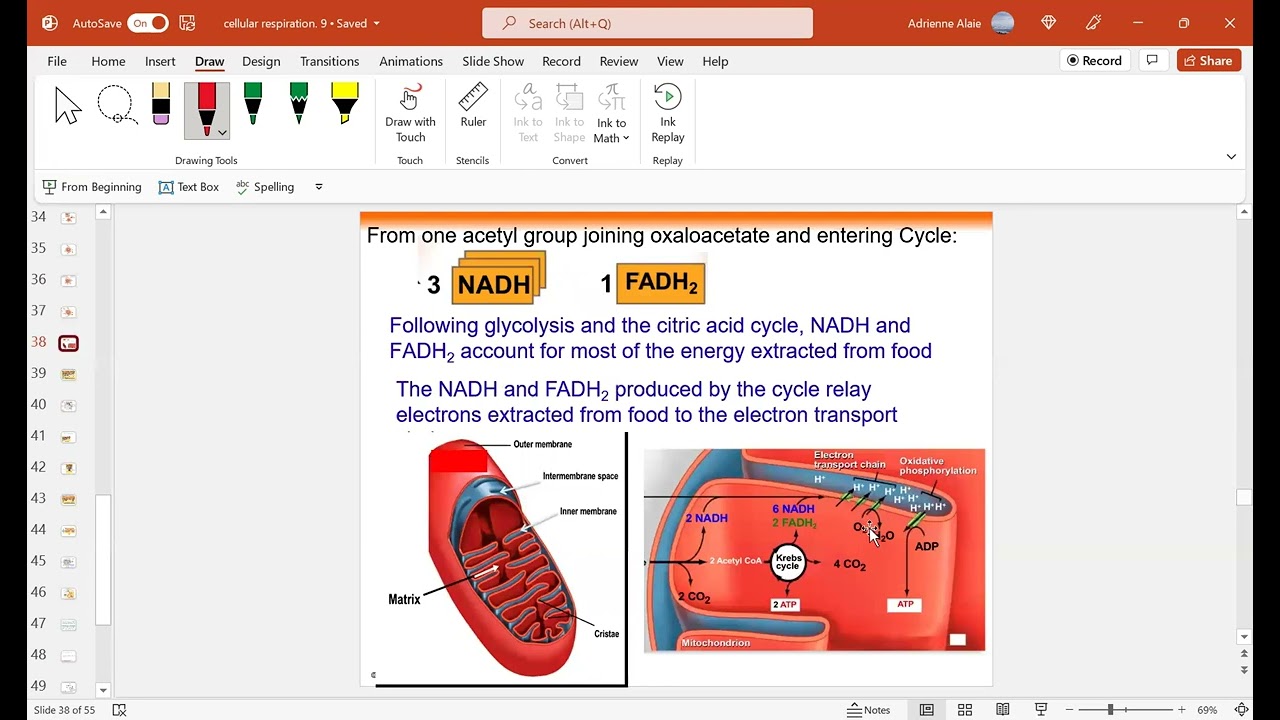
- Acetyl groups entering the Krebs cycle yield three NADH molecules and one FADH2 molecule per turn, representing significant energy extraction.
- NADH and FADH2 act as carriers for high-energy electrons derived from food breakdown, facilitating their transfer to the electron transport chain for ATP synthesis.
New Section
In this section, the discussion revolves around the electron transport chain and how NADH and FADH2 play a crucial role in linking glycolysis to cellular respiration.
Electron Transport Chain Process
- NADH and FADH2 carry energy from food sources, facilitating the transfer of electrons.
- Pyruvate oxidation in the matrix results in CO2 release and NAD+ reduction to form NADH.
- Acetyl groups from pyruvate enter the Krebs cycle, generating ATP, NADH, and FADH2.
- Remaining carbons from acetyl act as mobile carriers for electrons in the chain.
- Complexes 1 to 4 and mobile carriers like coenzyme Q facilitate electron transfer.
Specificity of Electron Flow
- Complexes 1 to 4 are labeled with multiple proteins for electron transfer specificity.
- Electrons flow specifically from NADH to complex 1 and FADH2 to complex 2 without short-circuiting.
- Oxidation-reduction reactions occur sequentially among complexes via mobile carriers like cytochrome c.
Energy Transfer Dynamics
- Complexes pass electrons through a series of reductions and oxidations until reaching oxygen as the final acceptor.
- The process is termed the electron transport chain due to sequential electron movement without short circuits.
Electron Transport Chain and ATP Synthesis
In this section, the discussion revolves around the electron transport chain, focusing on how electrons from NADH and FADH2 are utilized to generate a proton concentration gradient that drives ATP synthesis through chemiosmosis.
Electron Energy Transfer and Trapping
- Electrons from NADH and FADH2 undergo a stepwise transfer in the electron transport chain, leading to a drop in energy but crucially trapping this energy loss for further use.
Proton Pumping and Gradient Formation
- Electrons passing down the chain result in a decrease in free energy, ultimately being transferred to oxygen at its lowest energy state.
- Three complexes (Complex I, III, IV) facilitate proton pumping across the inner membrane with each NADH oxidized leading to about 10 protons being pumped.
Chemiosmosis and ATP Production
- The passage of electrons changes protein complexes' conformation, enabling them to pump protons against their concentration gradients.
- This establishes a proton concentration gradient across the inner membrane known as chemiosmosis, which is utilized to produce ATP through phosphorylation of ADP.
Role of ATP Synthase
- ATP synthase utilizes the proton concentration gradient by allowing protons to flow through it, driving phosphorylation for ATP generation.
- The enzyme uses exergonic flow of protons from higher to lower concentration for energetically favorable ATP production.
Efficiency of Energy Conversion
- For every four protons passing through ATP synthase, one molecule of ATP can be generated. This efficiency ties back to the initial oxidation of NADH and subsequent proton pumping processes.
New Section
In this section, the discussion revolves around the process of oxidative phosphorylation and the role of oxygen as the final electron acceptor in generating ATP.
Understanding Oxidative Phosphorylation
- Oxygen serves as the final electron acceptor in oxidative phosphorylation, leading to the phosphorylation of ADP to generate ATP.
- The electron transport chain facilitates the flow of electrons from one carrier to another, with each subsequent carrier having a higher affinity. This process allows for proton pumping and ultimately ATP production.
- Without oxygen, the electron transport chain would cease to operate, causing a backup of electrons in carriers like cytochrome c and disrupting ATP production.
- If there is no operational electron transport chain, NADH and FADH2 would be left holding electrons, halting their oxidation back to NAD+ and FAD essential for the citric acid cycle.
- The absence of oxygen leads to a halt in both the citric acid cycle and oxidative phosphorylation due to disrupted electron transfer processes.
New Section
This segment delves into how the absence of oxygen impacts the entire electron transport chain and its interconnectedness with ATP generation.
Impact on Electron Transport Chain
- Inadequate oxygen causes a backup in electrons along the electron transport chain, preventing NAD+ and FAD regeneration necessary for sustaining reactions in the citric acid cycle.
- The absence of oxygen disrupts both oxidative phosphorylation and citric acid cycle functions since oxygen acts as the final electron acceptor crucial for these processes.
New Section
Here, we explore how the electron transport chain sets up a proton gradient but does not directly produce ATP itself.
Role of Electron Transport Chain
- The primary function of the electron transport chain is to establish a proton concentration gradient rather than directly synthesizing ATP.