Clase 1 Fundamentos de absorción y conversión de composiciones
Absorption Process in Chemical Engineering
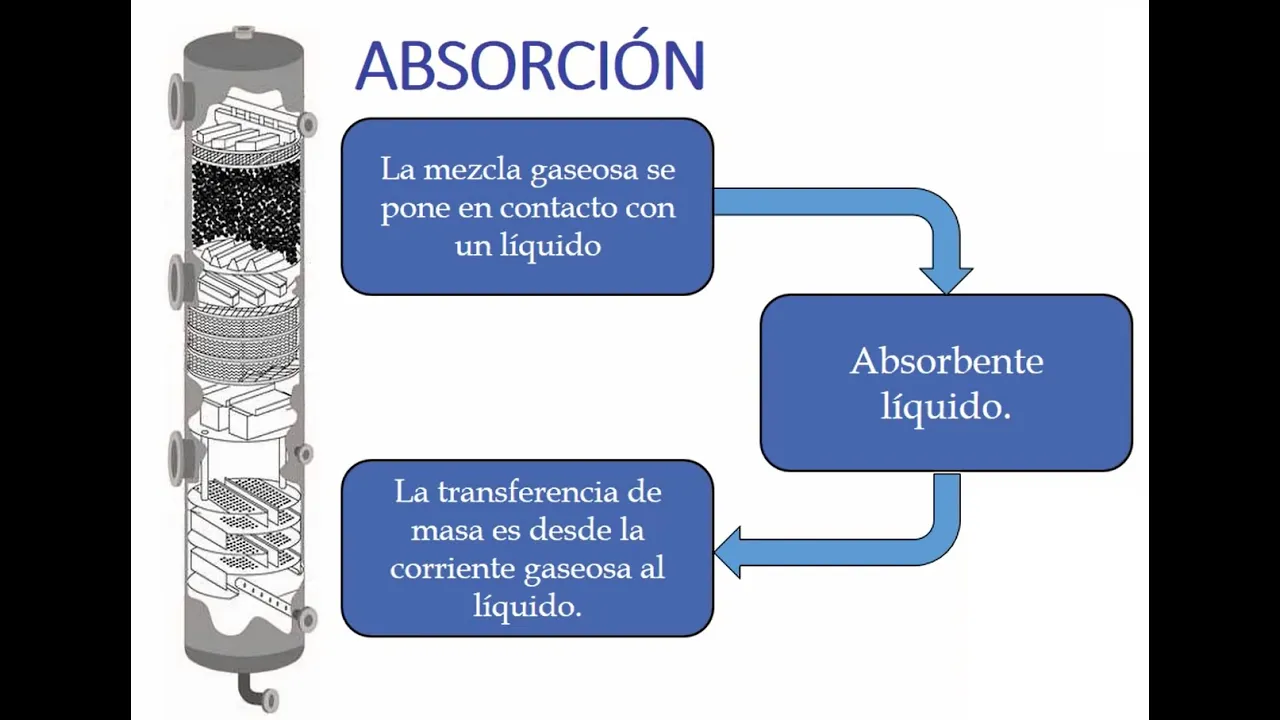
In this section, the concept of absorption in chemical engineering is discussed, focusing on the transfer of components between gas and liquid phases to form a solution.
Absorption Definition and Selective Dissolution
- Absorption is defined as the unit operation involving the selective dissolution of one or more gas components into a liquid phase.
Gas and Liquid Phase Compositions
- Absorption involves transferring components from the gas phase to the liquid phase based on their selectivity.
- The process considers compositions in both gas and liquid phases within an absorption column.
Transfer of Components and Ideal Conditions
- Transfer occurs when components move from gas to liquid phases, affecting the composition of each stream.
- The process aims for a rich liquid phase with high component concentration.
Moles Calculation and Temperature Consideration
- Moles calculation involves considering total moles in both gas and liquid phases.
- Temperature and pressure play crucial roles in absorption processes, impacting component transfer efficiency.
Fractional Molar Definition and Molar Ratio
This section delves into fractional molar concepts and molar ratios essential for understanding chemical engineering processes.
Fractional Molar Representation
- Fractional molar representation involves expressing component moles relative to total moles in a system.
Relationship Between Components
- Fractioanl molar values depict component proportions within a mixture accurately.
Molar Ratios Calculation
Understanding Molar Fraction and Molar Relationship
In this section, the speaker explains the concepts of molar fraction and molar relationship in a chemical context.
Moles of A and B
- The moles of A and B are determined by their respective fractions compared to the total moles.
- Moles of A over moles of B represent important ratios in chemical calculations.
Definitions and Calculations
- Key definitions include molar fraction and molar relationship for substances A and B.
- Calculation involves determining individual moles based on total moles and their respective fractions.
Calculating Molar Relationships
This part delves into calculating molar relationships using given molar fractions.
Molar Relationships
- Emphasizes distinguishing between molar fractions (lowercase letters) and molar relationships (uppercase letters).
- Explains how to derive expressions for molar relationships through total mole considerations.
Deriving Mole Ratios
The focus here is on deriving mole ratios from given expressions involving total moles.
Derivation Process
- Expressions for mole ratios are derived by manipulating equations involving total moles.
- Simplification leads to obtaining final expressions for mole ratios in terms of total moles.
Relating Mole Fractions to Mole Relationships
Connecting the concepts of mole fractions with mole relationships through mathematical transformations.
Mathematical Transformation
- Relates the relationship between molar fraction and molarity through a transformation equation.
- Introduces the concept of weight fraction as another parameter in chemical calculations.
Exploring Additional Equations in Chemical Calculations
Discussing further equations that enhance understanding in chemical calculations involving concentrations.
Further Equations
- Derives an equation linking molarity with weight fraction, expanding calculation possibilities.
Detailed Explanation of Gas Phase Calculations
In this section, the speaker explains calculations related to gas phase properties such as molar density and molecular weight.
Gas Phase Calculations
- The molar density of a component in the gas phase is determined by dividing its molar concentration by the total concentration of the gas phase.
- Total molar density is calculated by dividing the total pressure by the product of universal gas constant (R) and temperature (T).
- CTG and ROTEG are interchangeable notations used in these calculations for clarity.
- Given a mixture of CO2 and air with known molecular weights, identifying components based on their molecular weights is crucial for further calculations.
- By determining molecular weights, it becomes evident that CO2 represents compound A while air represents compound B in the given mixture.
Problem Solving: Gas Phase Calculations
This part delves into solving a problem involving a CO2-air mixture at specific conditions, calculating various parameters based on provided data.
Problem Solving Steps
- Analyzing a CO2-air mixture at defined temperature and pressure to calculate mole fractions and concentrations.
- Determining real molar ratio using mole fractions in gas phase components calculation.
- Computing weight fraction involves utilizing mole fractions and molecular weights of components A and B.
- Calculating weight ratios requires understanding binary systems' principles for accurate results.
Kelvin Conversion and Calculations
In this section, the speaker discusses Kelvin conversion rates and detailed calculations involving kilomoles, molecular weights, and total volumes.
Kelvin Conversion and Total Volume Calculations
- Kelvin conversion rate is 25 + 163.15 degrees per day.
- Total kilomoles can be canceled out in calculations to simplify the process.
- Calculation results in 5.76 x 10^-3 kg/mol for molecular weight over total cubic meters.
- The calculation involves multiplying the total moles by a specific value.
- Final calculation yields a value of 9.01 x 10^-5 kg/m^3.
Concentration Calculations
This part focuses on converting concentrations into percentages, parts per million, and milligrams per liter.
Concentration Conversions
- Conversion to percentage involves multiplying by 100.
- Parts per million (ppm) calculations are done by multiplying by 10^6.
- Parts per million in weight are determined similarly with a multiplication factor of 10^6.
- Final calculation results in a value of 6.315 for parts per million in weight.
Final Volume Calculations
The speaker delves into determining milligrams per liter of solution based on given values and conversions.
Determining Milligrams per Liter
- Conversion involves transforming kilograms to milligrams and cubic meters to liters.
- Calculation leads to obtaining a value of 5.76 mg/L for the solution volume.