The Basics of Organic Nomenclature: Crash Course Organic Chemistry #2
Crash Course Organic Chemistry: Understanding Nomenclature
The Importance of Language in Chemistry
- Deboki Chakravarti introduces the concept of language differences, using examples from everyday life to illustrate how terminology can vary across cultures.
- Highlights the complexities of language, particularly in chemistry, where different names may refer to the same substance (e.g., dichloromethane vs. methylene chloride).
- Discusses the establishment of IUPAC (International Union of Pure and Applied Chemistry) in 1919 to standardize chemical nomenclature for better global communication among chemists.
Historical Context of Chemical Naming
- By the time IUPAC was formed, organic chemistry had already been developing for over a century with many common names based on sources or characteristics (e.g., vanillin from vanilla beans).
- Common names often lack informative value regarding chemical structure, making systematic naming essential for clarity and understanding.
IUPAC Systematic Naming Rules
- Despite IUPAC's efforts, many common names persist due to their simplicity compared to systematic names like 4-hydroxy-3-methoxybenzaldehyde.
- Introduces three basic steps for systematic naming:
- Step 1: Identify the longest carbon chain and assign a root name.
- Step 2: Determine the highest priority functional group and add its suffix.
- Step 3: Identify substituents' types and positions on the carbon chain, adding numbered prefixes.
Carbon Chain Root Names
- Emphasizes counting carbons accurately to find the longest chain; arbitrary root names exist for chains up to four carbons (meth-, eth-, prop-, but-).
- For chains with five to twelve carbons, root names are derived from geometric shapes (e.g., pentagon = pent-, hexagon = hex-, etc.).
Types of Hydrocarbons
- Once a root name is established, a suffix indicates molecule type; hydrocarbons consist solely of hydrogen and carbon atoms.
- Discusses four types of hydrocarbons: alkanes (single bonds), alkenes (double bonds), alkynes (triple bonds), focusing primarily on alkanes, alkenes, and alkynes in this segment.
Characteristics of Alkenes and Alkynes
- Alkanes are described as stable compounds with single bonds that do not react easily; they are termed "low-energy couch potatoes."
- Alkenes feature double bonds which allow them to participate in more reactive processes compared to alkanes.
IUPAC Naming Rules for Organic Compounds
Understanding the Basics of IUPAC Naming
- The International Union of Pure and Applied Chemistry (IUPAC) establishes rules for naming organic compounds to ensure clear communication about molecular structures, particularly the location of double bonds.
- To name alkenes, identify the longest carbon chain, assign a root name based on its length (e.g., pent- for five carbons), and use the suffix -ene to indicate a double bond.
- Numbering starts from the end closest to the double bond; thus, a five-carbon chain with a double bond between carbons 1 and 2 is named pent-1-ene.
Incorporating Substituents in Naming
- Organic molecules often have substituents that replace hydrogen atoms. These are indicated by prefixes added before the root name, with numbering ensuring low values for substituent positions.
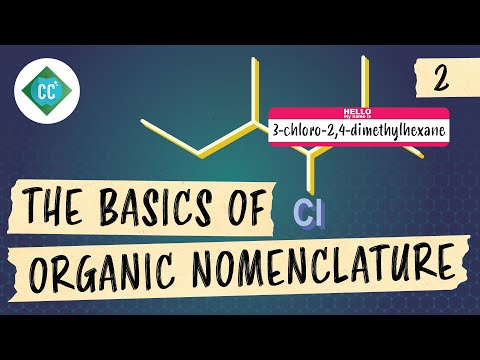
- For example, replacing one hydrogen in hexane with a -CH3 group results in 2-methylhexane. The prefix indicates both position and type of substituent.
Handling Multiple Substituents
- If multiple identical substituents exist, prefixes like di-, tri-, or tetra- are used. For instance, adding another -CH3 group leads to 2,4-dimethylhexane.
- When halogens replace hydrogens as substituents, their names change: chlorine becomes chloro-, bromine becomes bromo-, etc., which must also be included in alphabetical order when naming.
Advanced Naming Techniques
- In complex molecules with both alkenes/alkynes and various substituents, priority is given to functional groups over substituents during numbering.
- An example includes naming an alkene with an ethyl group at carbon 3 as 3-ethylpent-1-ene or an alkyne with two methyl groups as 2,7-dimethyloct-4-yne.
Finalizing Compound Names
- The process involves identifying the longest carbon chain (root), determining functional groups' priorities (alkenes vs. alkynes), and applying alphabetical order for all substituent prefixes.
- A compound like heptanediol can be named heptanone if it has ketone functional groups; however, when both types are present together (like bromine and methyl), they must be ordered correctly in the final name.
Practical Examples of IUPAC Names
- The complexity of names reflects their utility; e.g., "4-bromo-3-methylheptanone" provides precise information about structure rather than arbitrary names like "Debokiyne."
Common Names vs. IUPAC Names
Naming Molecules: Understanding the Structure
The Role of Chlorine in Molecular Structure
- The final structure of the compound is achieved by filling two open spots with chlorine, confirming that the lab partner's understanding was correct.
- Naming the compound as dichloromethane simplifies communication among scientists, ensuring clarity about the substance being discussed.
Steps to Naming Molecules
- There are three essential steps involved in naming molecules:
- Root name
- Suffix based on functional groups
- Prefix based on substituents