Que son los Espectros de emision y de absorcion
What Are Spectra?
Introduction to Continuous Spectrum
- The video begins by explaining that when light from a bulb passes through a quartz prism, it produces a continuous spectrum, displaying all colors and containing radiation of all wavelengths and frequencies.
- When the circuit is closed, the bulb emits white light which disperses into different frequencies or colors as it passes through the prism. This results in waves of varying energy levels, with red representing lower energy and violet indicating higher energy.
Emission Spectrum of Hydrogen
- The discussion shifts to what happens when hydrogen gas at low pressure is used instead of a white light bulb. The hydrogen emits a unique light characterized by specific lines in its spectrum.
- These emission lines represent distinct energies emitted by hydrogen atoms when excited (e.g., via an electric discharge). Each element has its own unique emission spectrum.
Comparison with Other Elements
- The speaker introduces helium as another example; upon excitation, helium shows completely different spectral lines compared to hydrogen.
- Neon is also mentioned as an example where its emission spectrum differs from both hydrogen and helium, emphasizing that each chemical element has a distinct emission spectrum.
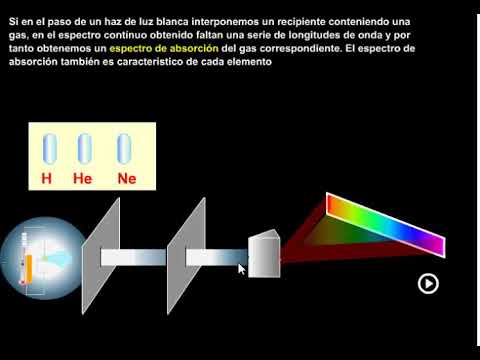
Absorption Spectrum Explained
- Transitioning to absorption spectra, the video explains how white light can be passed through a tube filled with low-pressure hydrogen. Instead of emitting light, the hydrogen absorbs certain wavelengths.
- As the white light passes through this medium, only specific colors are transmitted while others are absorbed, resulting in dark bands appearing on the spectrum—this is known as an absorption spectrum.
Characteristics of Absorption Spectra