Clasificación de compuestos orgánicos por estructura
Classification of Organic Compounds
Introduction to Organic Compounds
- The video introduces the classification of organic compounds, focusing on structural classification first, followed by functional group classification in a subsequent video.
Types of Organic Compounds
- Organic compounds can be classified into two main types: acyclic (open-chain) and cyclic (closed-chain). Acyclic compounds do not form geometric shapes, while cyclic compounds do.
Acyclic vs. Cyclic Compounds
- Acyclic compounds are characterized by open chains that start at one end and finish at another, whereas cyclic compounds form closed loops or geometric figures. This distinction is crucial for understanding their structure.
Zigzag Structure in Acyclic Compounds
- The zigzag structure represents acyclic compounds where each vertex corresponds to a carbon atom. For example, counting the vertices reveals the number of carbons present in the compound's structure.
Saturated vs. Unsaturated Compounds
- The classification further divides organic compounds into saturated and unsaturated based on bond types:
- Saturated compounds have only single bonds and are fully bonded with hydrogen atoms.
- Unsaturated compounds contain double or triple bonds, allowing for additional hydrogen atoms to attach if those bonds are broken.
Linear vs. Branched Structures
- Lastly, acyclic compounds can be linear or branched:
- Linear structures follow a straight chain without deviations.
- Branched structures have side chains extending from the main carbon chain, indicating complexity in their arrangement.
Examples of Acyclic Compounds
- Three examples of acyclic compounds are presented; all begin at one point and end at another without forming cycles, confirming their classification as acyclic. This reinforces the concept of general-to-specific classifications within organic chemistry.
Final Classifications Recap
- To summarize:
- Identify if a compound is cyclic or acyclic.
- Determine if it is saturated (single bonds) or unsaturated (double/triple bonds).
- Classify as linear or branched based on structural arrangement.
This systematic approach aids in understanding organic compound classifications effectively.
Classifications of Cyclic Compounds
Overview of Saturated and Unsaturated Compounds
- The discussion begins with the classification of compounds as saturated, emphasizing their simple linkages.
- A distinction is made between branched and linear structures, noting that a compound can appear branched while still following a linear carbon chain.
- The importance of carbon continuity in determining whether a compound is classified as linear is highlighted.
Characteristics of Cyclic Compounds
- Cyclic compounds are defined by their ability to form closed loops; they can be saturated or unsaturated based on the types of bonds present.
- The presence of double or triple bonds indicates that a compound is unsaturated, allowing for additional hydrogen atoms if these bonds are broken.
Types of Cyclic Structures
- Three classifications for cyclic compounds are introduced: branched vs. linear, saturated vs. unsaturated, and simple vs. branched.
- An explanation follows regarding how cyclic compounds can form geometric shapes through their structure.
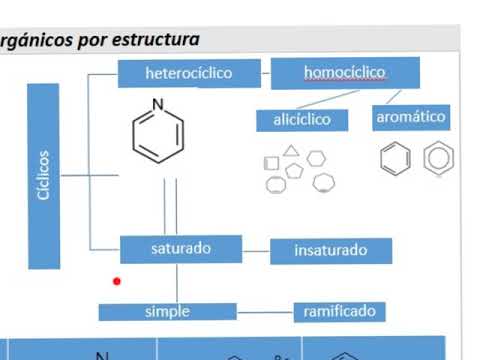
Heterocyclic vs. Homocyclic Compounds
- Four classifications for cyclic compounds include heterocyclic (containing elements other than carbon at vertices), homocyclic (all vertices occupied by carbon), saturated/unsaturated, and simple/branched.
- The definition of hetero versus homocyclic depends on the presence of non-carbon elements at the vertices.
Detailed Classification Insights
- If any vertex contains an element other than carbon (e.g., nitrogen), it qualifies as heterocyclic; otherwise, it remains homocyclic.
- Examples illustrate how substituting different elements like oxygen or sulfur changes the classification from homocyclic to heterocyclic.
Aromatic vs. Aliphatic Homocycles
- Within homocyclic compounds, further distinctions arise: aromatic (like benzene with alternating double bonds in a hexagonal shape) versus alicyclic (which do not contain benzene).
- Aromatic compounds must contain specific structural features such as those found in benzene; anything lacking this structure falls under alicyclic.
Summary of Classifications
- The primary classifications discussed include cyclic nature, whether they are hetero or homo cíclicos, saturation levels, and branching characteristics.
- Emphasis is placed on understanding these classifications to better analyze chemical structures and their properties within organic chemistry contexts.
Classification of Cyclic Compounds
Overview of Cyclic Compounds
- The speaker introduces the classification of compounds, specifically focusing on cyclic compounds, which form cycles.
- The first classification is identified as "heterocyclic" due to the presence of nitrogen in the compound, indicating a break in the chain.
Types of Cyclic Compounds
- The second classification addresses whether the compound is saturated or unsaturated; this particular compound has a double bond, making it unsaturated.
- The third classification involves determining if the compound is simple or branched. This specific compound is classified as simple.
Further Classifications and Examples
- A new cyclic compound is introduced; it appears heterocyclic but does not have interruptions at its vertices, confirming it as homocyclic.
- Both discussed compounds are homocyclic; however, one must determine if they are aromatic or aliphatic.
Aromatic vs Aliphatic Compounds
- The distinction between aromatic and aliphatic compounds is made based on their structure. One example contains benzene (aromatic), while another does not have sufficient double bonds to be classified as aromatic.
- Benzene's structure with three double bonds confirms its aromatic nature compared to other non-aromatic cyclic structures.
Final Classifications and Summary
- Both examples are confirmed as cyclic; one being alicyclic (non-aromatic), while the other retains its aromatic classification due to benzene content.
- Both compounds are also classified as unsaturated due to their double bonds present in their structures.
- A final comparison shows that both examples exhibit branching characteristics, leading them to be categorized as branched compounds.
Conclusion on Compound Classifications
- The discussion concludes with a summary that cyclic compounds have four classifications while acyclic ones only possess three classifications.
- An overview table will summarize these classifications for better understanding and preparation for exams.