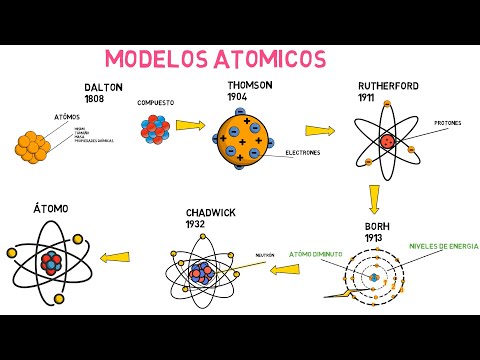
Modelos atómicos (Dalton, Thomson, Rutherford, Bohr y Chadwick)
New Section
The evolution of the atomic theory from Democritus to Chadwick, highlighting key discoveries and models that shaped our understanding of the atom.
Democritus and John Dalton
- Atoms as indivisible solid particles -
- Dalton's precise definition of atoms in 1808 -
- Elements composed of identical atoms with unique properties -
Joseph Thompson and Ernest Rutherford
- Thompson's discovery of electrons within the atom -
- Rutherford's gold foil experiment revealing nucleus and electrons' distribution -
Niels Bohr
- Bohr's planetary model introducing energy levels for electrons -
- Electrons transitioning between energy levels emitting or absorbing photons -
James Chadwick
- Chadwick's discovery of neutrons resolving atom weight discrepancies -