4 - Linfocitos T
Understanding T Lymphocytes and Their Development
Overview of T Lymphocyte Generation
- The seminar focuses on T lymphocytes, covering their generation, activation requirements, immune response profiles, and memory.
- The T cell receptor (TCR) is formed during ontogeny before antigen exposure through a process called somatic recombination.
Structure of the T Cell Receptor (TCR)
- The TCR consists of alpha and beta chains; the variable region of the beta chain shows significant variability compared to the more conserved carboxy-terminal regions.
- The variable portion of the beta chain is illustrated in three colors to represent its diverse generation through genetic rearrangement.
Genetic Rearrangement Process
- In non-T lymphocyte cells and precursor T cells, genetic material exists in a germline configuration until somatic recombination occurs.
- A specific fragment (B1 from J segments) undergoes rearrangement, leading to a new beta chain with a variable region after transcription and translation.
Formation of Alpha Chain
- The alpha chain's variable region is generated by combining two fragments known as B&J segments found in all precursor cells.
- Enzymes called recombinases recognize these genetic fragments, cut them, and other enzymes join them together to form functional receptors.
Mechanisms for Diversity Generation
- Somatic recombination allows for an extensive variety of B cell receptors (BCR), contributing to antigenic diversity due to imprecise joining mechanisms.
- This diversity arises not only from different configurations but also from errors introduced during the joining process that affect nucleotide sequences.
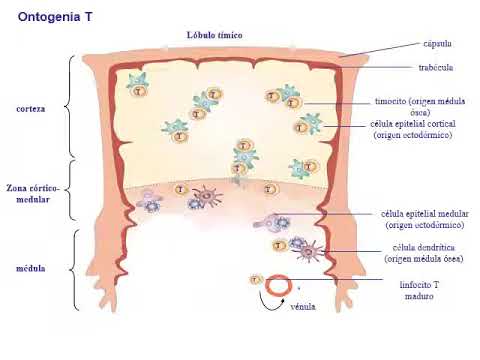
Ontogeny in Thymus
- Once successfully formed, the open-chain structure expresses on the membrane alongside a surrogate alpha chain; this triggers cellular proliferation upon successful arrangement.
- Each daughter cell inherits this arrangement while further diversifying through additional combinations with various alpha chains generated during thymic development.
Importance of Precise Genetic Rearrangement
- The specificity of receptors comes from how well they can bind antigens; thus, variations in nucleotide sequences enhance recognition capabilities.
- Even if identical fragments are chosen for rearrangement, imprecise joining leads to unique nucleotide sequences that contribute to receptor diversity.
Migration and Training within Thymus
- Hematopoietic stem cells migrate from bone marrow to thymus where crucial steps in lymphocyte development occur within distinct thymic lobules.
T-Cell Ontogeny and Thymocyte Development
Thymocyte Environment and Initial Stages
- Thymocytes encounter a unique environment in the thymic cortex, primarily composed of cortical thymic epithelial cells, differing from those found in the medullary region.
- The development of T lymphocytes involves specific markers; thymocytes are categorized based on co-receptor expression (CD4 and CD8). Those lacking both are termed double-negative cells.
Progression to Positive Selection
- During T-cell ontogeny, thymocytes transition from double-negative to double-positive states as they begin expressing co-receptors, ultimately maturing into single-positive T lymphocytes.
- Initially, double-negative thymocytes proliferate rapidly within the cortical zone without expressing CD4 or CD8. This stage is crucial for defining their lineage.
Rearrangement of TCR Genes
- The majority of circulating T lymphocytes undergo significant rearrangements in their beta chain genes (V(D)J recombination), which is essential for functional T-cell receptor (TCR) formation.
- Successful rearrangement leads to transcription and translation of the beta chain, allowing it to pair with a surrogate alpha chain on the thymocyte surface.
Positive Selection Mechanism
- Once both chains are expressed on the cell membrane, signals trigger proliferation and further maturation. This process marks a critical checkpoint in T-cell development.
- A productive rearrangement results in a functional TCR that must interact with major histocompatibility complex (MHC) molecules presented by cortical epithelial cells for survival.
Central Tolerance Induction
- The interaction between double-positive thymocytes and MHC molecules is vital; failure to engage leads to apoptosis through a process known as "death by neglect."
- Positive selection occurs when thymocytes successfully recognize self-MHC molecules, determining whether they will express CD4 or CD8 based on their interactions during this phase.
Final Stages of Development
- Only a subset of thymocytes survives positive selection; these selected cells migrate towards medullary regions where further maturation occurs.
T Cell Development and Activation
Thymocyte Migration and Selection Processes
- Thymocytes migrate from the thymic cortex to the medulla, where they express various tissue-specific proteins. The transcription factor Aire is crucial for regulating genes expressed in other tissues.
- Thymocytes that receive strong signals through their TCR (T Cell Receptor) are deemed autoreactive and undergo negative selection, leading to cell death.
- A double-positive thymocyte must be positively selected in the cortex before migrating to the medullary zone, successfully passing through negative selection without receiving intense signals.
- Surviving thymocytes exit the thymus as mature single-positive T cells, capable of recognizing self-MHC molecules specific to each individual.
- Not all self-proteins enter the medullary zone; autoreactive T cells are controlled by mechanisms known as peripheral tolerance induction.
Ontogeny of Lymphocytes
- The mechanisms involved in lymphocyte ontogeny include positive and negative selection processes that allow T cells to emerge with antigen receptors for unknown antigens.
- Mature naïve T cells traffic through secondary lymphoid organs, where only professional antigen-presenting cells can activate them upon encountering their specific antigens.
Dendritic Cell Activation and Function
- Immature dendritic cells in tissues can be activated by pathogen-associated molecular patterns (PAMPs) and pro-inflammatory cytokines, enhancing their ability to present antigens.
- Activated dendritic cells express chemokine receptors that guide them via lymphatic vessels to secondary lymphoid organs, positioning themselves strategically within these areas for optimal interaction with naïve T cells.
Interaction Between Naïve T Cells and Dendritic Cells
- Naïve T cells entering secondary lymphoid organs search for specific peptide-MHC complexes on dendritic cells; if not found, they will leave the node and continue circulating.
- Upon finding a matching complex on a dendritic cell's surface, naive T cells initiate activation processes which involve multiple signaling pathways leading to proliferation and differentiation.
Requirements for Naïve T Cell Activation
- For activation, naïve T cells require two signals: recognition of peptide-MHC complexes by their TCR (first signal), followed by co-stimulatory signals involving CD28 interacting with CD80/CD86 on mature dendritic cells (second signal).
- Successful reception of both signals stabilizes IL-2 messenger RNA production; activated naïve T cells begin producing IL-2 while expressing high-affinity IL-2 receptors necessary for further proliferation.
T-Cell Activation and Dendritic Cell Interaction
Clonal Expansion of T-Cells
- The process begins with high affinity clonal expansion of T-cells due to IL-2 production, which is a result of signals received from dendritic cells.
Reciprocal Activation Between Dendritic Cells and T-Cells
- Activated T-cells express CD40L, interacting with CD40 on dendritic cells, enhancing the latter's ability to stimulate T-cell responses.
Co-stimulation Requirements for T-Cell Activation
- CD4+ and CD8+ naïve T-cells require recognition of MHC-peptide complexes along with co-stimulatory signals for activation; however, CD8+ T-cells need higher levels of these signals compared to CD4+.
Importance of Dendritic Cell Interactions
- Naïve CD8+ T-cells often depend on prior interaction between dendritic cells and activated CD4+ T-cells to enhance their co-stimulatory capacity for effective activation.
Consequences of Inadequate Co-stimulation
- If naïve T-cells fail to receive adequate co-stimulatory signals after recognizing antigenic peptides, they become anergic—losing the ability to activate and eventually undergoing apoptosis.
Migration Patterns of Activated T-Cells
Chemokine Receptor Expression in Naïve vs. Activated T-Cells
- Naïve lymphocytes migrate through secondary lymphoid organs due to specific chemokine receptors; upon activation, they alter adhesion molecule expression patterns allowing access to peripheral tissues.
Role of Dendritic Cells in Tissue-Specific Responses
- The timing and location of naïve T-cell activation by dendritic cells influence their migration profile towards specific tissues based on released substances during activation.
Differentiation into Effector Cells
Influence of Tissue-Derived Signals on Effector Functionality
- For instance, intestinal dendritic cells release retinoic acid that prompts effector or memory T-cells to express specific adhesion molecules facilitating access to the intestinal mucosa.
Skin-Derived Signals Affecting Memory Cell Migration
- Conversely, skin-derived dendritic cells produce calcitriol (active Vitamin D3), influencing effector memory cell patterns for accessing skin tissue post activation.
Mechanisms of Cytotoxicity in Effector T-Cells
Activation Timeline Post Antigen Encounter
- The complete process from activation through clonal expansion and differentiation into cytotoxic effector cells takes approximately five days following antigen exposure.
Targeting Infected Cells
- Once activated, cytotoxic effector T-cells seek out infected host cells using adhesion molecules like LFA1 for effective targeting based on previous encounters with antigens presented by dendritic cells.
Mechanisms of Cytotoxic T Cells and Their Activation
Cytotoxic Functions of CD8 T Lymphocytes
- CD8 cytotoxic T lymphocytes can perform effector functions such as producing pro-inflammatory cytokines and inducing apoptosis in target cells upon receiving activation signal 1.
- There are two cytotoxic mechanisms employed by CD8 T lymphocytes: a non-secretory mechanism involving direct interaction with target cells, and a secretory mechanism that releases perforins and granzymes.
Comparison with Natural Killer (NK) Cells
- Unlike NK cells, which activate based on a balance of activating and inhibitory signals, CD8 T lymphocytes recognize specific antigens presented on target cells to initiate their cytotoxic response.
- Upon recognizing the antigenic complex on the target cell, activated CD8 T lymphocytes receive activation signal 1, leading to the induction of apoptosis in the target cell.
Differentiation Pathways for Naive CD4 T Lymphocytes
- Naive CD4 T lymphocytes have significant potential to differentiate into various effector profiles depending on cytokine environments encountered during their activation.
- The differentiation process is influenced by interactions with dendritic cells in secondary lymphoid organs, where cytokine presence determines whether they become Th1 or Th17 effector cells.
Profiles of Effector CD4 T Lymphocytes
- Th1 and Th17 profiles are characterized by specific cytokine production; Th1 produces interferon-gamma while Th17 is involved in responses against bacterial and fungal infections.
- The role of Th1 cells includes immunity against intracellular microorganisms and antiviral responses.
Factors Influencing Differentiation into Various Subtypes
- The presence of IL-21 during naive CD4 T cell activation favors differentiation towards follicular helper T (Tfh) cells, which assist B cell activation.
- In contrast, high levels of IL-4 promote differentiation towards the Th2 profile, which is crucial for combating helminth parasites and allergic diseases.
Regulatory Mechanisms in Immune Response
- Naive CD4 T lymphocyte differentiation can also lead to regulatory profiles under certain conditions; IL-10 promotes regulatory type 1 (Tr1), while transforming growth factor-beta (TGF-beta) leads to Tr3 regulatory cells.
- These regulatory subsets play essential roles in modulating immune responses to prevent overactivity that could lead to autoimmunity.
Complexity of Immune Responses
- During immune challenges like infections or tumors, multiple effector profiles may coexist; for instance, both Th1 responses and antibody production can occur simultaneously.
- Cytokines produced by one type of effector cell can inhibit the differentiation pathways of others; for example, IL-4 inhibits Th1 development while interferon-gamma suppresses Th2 differentiation.
Transcription Factors Governing Differentiation
- Different transcription factors regulate the expression patterns associated with each effector profile. For instance:
- Th1 requires transcription factors like TBET and STAT4 for interferon-gamma stimulation.
- Th2 differentiation involves GATA3 suppression through these same factors.
Inflammatory Response and Immune Activation
Role of Macrophages in Inflammation
- Activated macrophages play a crucial role in mediating inflammatory responses in peripheral tissues, with interferon gamma and IL-2 being key factors that activate NK cells and promote CD8 T cell activation.
Mycobacterium tuberculosis and Tuberculosis
- Mycobacterium tuberculosis is the causative agent of tuberculosis, primarily affecting the lungs but can also impact other organs. It spreads through airborne bacteria expelled by infected individuals during coughing.
- According to a 2019 WHO report, tuberculosis is one of the top ten causes of mortality worldwide, with approximately one-quarter of the global population infected.
Immune Response to Mycobacterial Infection
- Upon reaching pulmonary alveoli, macrophages and dendritic cells ingest mycobacteria, leading to the release of pro-inflammatory cytokines that recruit more leukocytes.
- Specific Th1 lymphocytes activate macrophages to control mycobacterial growth within granulomas, which contain the pathogen effectively for years.
Granuloma Formation and Latent Infection
- Granulomas form as a containment strategy against mycobacteria; however, if immune surveillance weakens (e.g., due to corticosteroids), granulomas may liquefy, releasing bacilli into airways.
Th17 Cells and Their Functions
Role in Autoimmunity and Extracellular Infections
- Th17 cells are central not only in autoimmune phenomena but also in immunity against extracellular bacterial infections and fungi.
Th2 Cells: Key Players Against Parasites
Collaboration with B Lymphocytes
- Follicular helper T cells collaborate with naïve B lymphocytes by providing secondary activation signals essential for humoral responses.
Immune Response Against Allergens
- Th2 cells are critical for immune responses against extracellular parasites; they induce mast cell degranulation and stimulate mucus secretion to expel parasites.
Cytokine Production by Th2 Cells
Mechanisms Supporting Antibody Production
- Cytokines produced by Th2 cells facilitate antibody class switching towards IgE production specific for pathogens, enhancing opsonization for destruction via antibody-dependent cellular cytotoxicity.
Tissue Repair and Allergy Development
Understanding Regulatory T Cells and Immunological Memory
Role of Regulatory T Cells
- The discussion begins with the role of Th2-type responses, highlighting the reproduction of mast cells and the functions of regulatory T cells (Tregs) in maintaining homeostasis and tolerance by limiting autoreactive clones in peripheral tissues.
- Natural regulatory T cells are identified as CD4+ Foxp3+ lymphocytes that migrate directly from the thymus, possessing regulatory capabilities. Inducible regulatory T cells, on the other hand, differentiate from CD4+ T cells upon recognizing antigens in secondary lymphoid organs.
Immune Response Dynamics
- The seminar focuses on understanding immunological memory generation during primary infections, illustrating phases such as innate immune response induction and adaptive immune response development leading to effector T cell generation.
- It is emphasized that immunological memory ensures a faster and more efficient response upon re-exposure to the same pathogen. A graph illustrates how specific T cell numbers increase after initial exposure.
Memory Cell Generation
- Following an initial antigen challenge, effector lymphocytes peak before undergoing contraction; remaining cells differentiate into memory T cells. This process enhances future responses to subsequent encounters with the same pathogen.
- The importance of generating immunological memory is reiterated, showing how repeated exposures can lead to increased populations of memory T cells capable of rapid activation and differentiation into effector cells when encountering pathogens again.
Activation Mechanisms
- Memory T cells can proliferate in response to pro-inflammatory cytokines like IL-12 or type I interferons even in the absence of their specific antigen. This highlights their adaptability during various infections.
- Two main types of memory T cells are discussed: central memory (similar to naive lymphocytes with L-selectin expression for migration) and effector memory (lacking L-selectin but resembling effector T cell migration patterns).
Resident Memory Cells
- Effector resident memory T cells can establish stable populations within tissues, acting as sentinels against reinfection. Their ability to respond quickly upon re-exposure is crucial for effective immunity.
- The dynamics between activated memory T cells and their roles during reinfection are explored, emphasizing how they reside in tissues ready to respond rapidly without needing full systemic activation.