Bioelementos
Bioelements and Their Importance in Human Health
Introduction to Bioelements
- The class focuses on bioelements, which are organized in a periodic table. Some heavy metals like arsenic, lead, and mercury are toxic to human health.
- The bioelements discussed are those found naturally in living beings, particularly humans.
Primary Bioelements
- Out of all bioelements present in humans, only four (oxygen, carbon, hydrogen, nitrogen) make up over 98% of the body composition:
- Oxygen: 65%
- Carbon: ~18%
- Hydrogen: ~10%
- Nitrogen: ~3%
- Oxygen, carbon, and hydrogen are essential components of major biomolecules such as carbohydrates, lipids, and proteins. Nitrogen is also crucial for proteins and nucleic acids.
Secondary Bioelements
- Other bioelements exist in smaller proportions but are vital for metabolism. They include:
- Calcium (1.5%)
- Phosphorus (1%)
Classification of Bioelements
- Bioelements can be classified into three groups based on their abundance:
- Primary: Carbon, hydrogen, oxygen, nitrogen, phosphorus, sulfur.
- Secondary: Sodium, potassium, calcium, magnesium, chlorine.
- Oligoelements: Iron, zinc, copper, iodine, selenium.
Functions of Each Group
- Primary bioelements constitute more than 95% of an organism's composition; secondary elements form about 4-5%, primarily forming soluble or insoluble salts.
- Oligoelements are present in very small amounts (<1%) but play critical roles in bodily functions.
Detailed Look at Key Elements
- Carbon (C), hydrogen (H), and oxygen (O) are ubiquitous in consumed molecules like carbohydrates and proteins.
- Nitrogen (N), abundant in the air (~78%), is part of amino acids and nucleic acids but cannot be directly metabolized by humans.
Nitrogen Fixation Process
- Nitrogen fixation is as crucial as photosynthesis; it involves bacteria that convert atmospheric nitrogen into usable forms for plants.
- Rhizobacteria create nodules on leguminous plant roots to absorb nitrogen from the air while providing carbon sources back to the bacteria—a mutualistic relationship.
Aquatic Organisms Involved in Nitrogen Fixation
- In aquatic environments like lagoons or freshwater bodies live organisms called Nostoc that also fix nitrogen through specialized cells known as heterocysts.
Characteristics of Cianobacterias
- Cyanobacteria (formerly known as blue-green algae), which fix atmospheric nitrogen efficiently. They have significant implications for protein production due to their ability to utilize atmospheric nitrogen effectively.
Bioelements and Their Functions in the Body
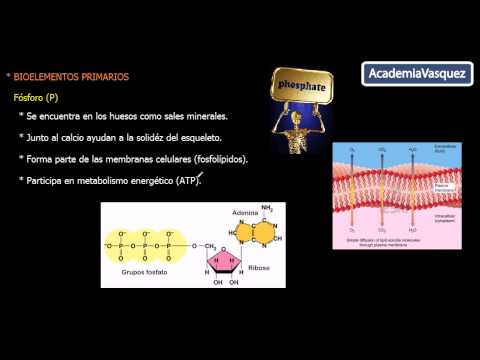
Primary Bioelements: Phosphorus and Sulfur
- Phosphorus is found in bones and teeth as mineral salts, contributing to skeletal solidity through phosphates.
- It is a key component of phospholipids in cell membranes, which facilitate nutrient intake and waste elimination.
- Phosphorus is crucial for energy metabolism, forming part of adenosine triphosphate (ATP), essential for cellular functions.
- Sulfur, represented by 'S', is integral to two amino acids—methionine and cysteine—that are vital for protein formation.
- Additionally, sulfur contributes to B vitamins like thiamine (B1) and biotin (B8), as well as collagen synthesis, which provides skin stability.
Secondary Bioelements: Sodium, Potassium, Magnesium, Calcium
Sodium and Potassium
- Secondary bioelements constitute 4-5% of cellular composition; they form mineral salts that can be soluble or insoluble in water.
- Sodium (Na) and potassium (K) play critical roles in maintaining cellular balance; their levels inversely affect each other.
- These ions regulate water movement into/out of cells; increased sodium leads to decreased potassium levels.
Magnesium
- Magnesium (Mg), essential for chlorophyll production in plants, also aids bone density by facilitating calcium absorption.
- It serves multiple roles including acting as an antacid or laxative due to its presence in products like magnesium hydroxide.
Calcium
- Calcium (Ca), making up 99% of bone structure, provides rigidity to bones and teeth while also aiding enzyme function.
- It plays a role in blood pressure regulation and muscle contraction; deficiencies can lead to conditions such as rickets in children or osteoporosis in adults.
Importance of Trace Elements in Metabolism
Overview of Trace Elements
- The discussion begins with the significance of trace elements, which are present in very low proportions (less than 1%) but are essential for metabolic functions.
Iron (Fe)
- Iron is a crucial component of hemoglobin, the protein responsible for oxygen transport in red blood cells. A deficiency leads to anemia, causing fatigue and reduced performance.
- Hemoglobin's structure centers around iron, highlighting its role in effective oxygen delivery throughout the body.
Zinc (Zn)
- Zinc plays a vital role in strengthening the immune system and is involved in hair and nail development. It helps prevent stress and fatigue through dietary intake.
Copper (Cu)
- Copper exists as ions and is integral to various enzymes that support immune, nervous, and cardiovascular systems. It also aids in red blood cell formation but to a lesser extent than iron.
Iodine (I)
- Iodine is essential for thyroid function; its deficiency can lead to goiter and impaired growth. Sources include marine foods and iodized salt.
Cobalt (Co)
- Cobalt is found in vitamin B12 (cobalamin), which supports red blood cell production and maintains central nervous system health.
Selenium (Se)
- Selenium contributes to reducing cellular aging by forming antioxidant enzymes that combat free radicals linked to aging processes. Research suggests it may help prevent cancer.
Manganese (Mn)
- Manganese is important for newborn growth, bone formation, and acts as an enzyme activator that enhances metabolic reactions by increasing product yield over time when combined with enzymes.
Fluorine (F)
Bioelements and Their Importance in Living Organisms
Overview of Bioelements
- Bioelements are essential chemical elements that constitute all living beings. They can be classified based on their composition rather than their importance, as all are crucial for life.
Primary Bioelements
- The primary bioelements make up over 95% of the total composition in living organisms. This group includes Carbon (C), Hydrogen (H), Oxygen (O), Nitrogen (N), Phosphorus (P), and Sulfur (S).
- While P and S are present in smaller proportions compared to C, H, O, and N, they play vital roles in important structures such as cell membranes (e.g., phospholipids) and proteins.
Secondary Bioelements
- Secondary bioelements account for about 4-5% of the total composition. They include Sodium (Na), Potassium (K), Calcium (Ca), Magnesium (Mg), and Chlorine (Cl). These elements often form soluble or insoluble salts.
Trace Elements