Episódio 13 - Modelando os Elétrons #2 (Por que os orbitais tem esses formatos? #2)
New Section
In this section, the speaker introduces the topic of modeling electrons in chemistry, focusing on the angular and radial components of wave functions.
Modeling Electrons in Chemistry
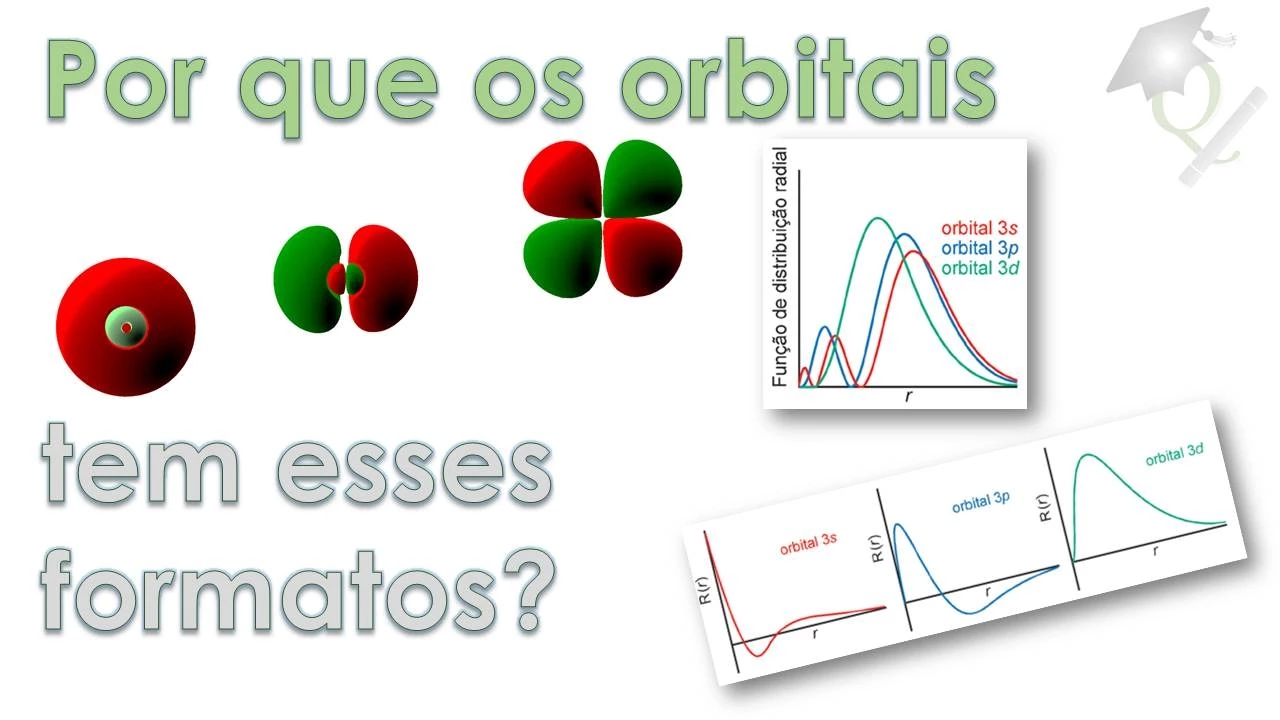
- The angular part of the wave function determines the shape of orbitals like s, p, d.
- The radial part of the wave function influences the size or extension of orbitals and energy levels.
- Radial functions differ for various orbitals (e.g., 1s, 2s) impacting energy levels and nodal regions.
- Radial functions exhibit exponential decay towards zero at large distances from the nucleus.
- Orbitals with different angular momentum quantum numbers have distinct radial behaviors.
Understanding Orbital Shapes and Nodes
In this section, the speaker delves into the relationship between variables in R and x within a second-degree function. The discussion progresses to exploring how to determine the roots of a second-degree function using the quadratic formula, leading to insights on orbital shapes and nodal regions.
Relationship Between Variables in Second-Degree Functions
- Understanding the transition from R to x in a variable like R reveals a second-degree function.
- By applying the quadratic formula to this polynomial, one can ascertain two values of R that define the function's behavior.
Orbital Shapes and Nodal Regions
- Orbital shapes are elucidated through visualizing spheres before and after nodal regions.
- The radial nodal regions increase with higher energy levels; for instance, an orbital 1s lacks nodal regions while an orbital 3s has two radial nodal regions.
- Emphasizing the significance of squared wave functions for determining electron probability density at varying distances from the nucleus.
Correlation Between Bohr Model and Quantum Model
This segment explores parallels between Bohr's deterministic model and quantum mechanics concerning electron distribution within atoms, highlighting similarities without equating both models.
Parallels Between Bohr Model and Quantum Mechanics
- Drawing comparisons between Bohr's model indicating electron position at 0.529 angstrom with quantum mechanics' highest electron density at the same distance.
- Acknowledging similarities as mere resemblances rather than identical models or reductions of one another.
Diverse Characteristics of Different Orbitals
The discussion shifts towards analyzing distinct characteristics of various orbitals such as s, p, and d types, focusing on their unique features including nodal patterns.
Analysis of Orbital Characteristics
- Detailed examination of s-type orbitals showcasing second-degree polynomials resulting in spherical shapes with radial nodal regions.
- Contrasting s-orbitals with p-orbitals characterized by different curve formats denoting angular nodes alongside radial nodes.
Nodal Patterns in Different Orbitals
This part delves into intricate details regarding nodal patterns within different orbitals like P and D types, emphasizing their distinctive properties.
Nodal Patterns in P-Type Orbitals
- Highlighting singular angular node presence in P-orbitals alongside radial nodes influencing their structure.
Nodal Patterns in D-Type Orbitals
- Illustrating trivial zero points for D-orbitals at specific radii signifying unique structural characteristics devoid of additional radial nodes except for trivial ones at R = 0.
Understanding Nodal Regions in Orbitals
In this section, the speaker discusses the relationship between angular and radial nodal regions in orbitals, focusing on the example of the 3pz orbital.
Relationship Between Angular and Radial Nodal Regions
- The angular nodal region corresponds to the angular origin node, while the radial nodal region separates parts of the function as a radial origin node.
- Orbitals like 3P have two nodal regions: one angular and one radial. The total number of nodal regions is given by n - 1, where n is the energy level.
- Secondary quantum number (L) corresponds to the number of angular nodal regions. For instance, L for S is 0, for P is 1, and for D is 2. The remaining nodal regions are radial.
Examples of Nodal Regions in Different Orbitals
- The 5S orbital has four total nodal regions with all being radial. In contrast, the 4P orbital has three nodal regions with one being angular.
- The 6D orbital exhibits five total nodal regions with two being angular and three being radial. Angular nodes take precedence in defining the orbital characteristics over radial nodes.
Analyzing Total Nodal Regions in Orbitals
This section delves into determining the total number of angular and radial nodal regions in various orbitals based on their quantum numbers.
Determining Nodal Regions Based on Quantum Numbers