Collagen protein
What is Collagen and Why is it Important?
Overview of Collagen
- Collagen is the most important fibrous protein in animal cells, particularly in mammals, and is essential for connective tissues.
- It plays a crucial role in various structures including bones, teeth, cartilage, tendons, ligaments, skin, and blood vessels.
Abundance and Composition
- Collagen constitutes about 30% of the total protein mass in vertebrates, making it the most abundant protein in their bodies.
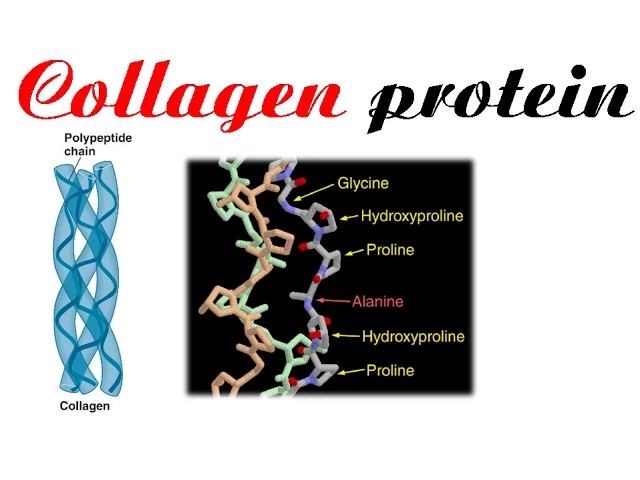
- The general structure of collagen is characterized as a triple helical protein that can withstand significant tensile strength.
Amino Acid Composition
- Collagen has a unique amino acid composition with three repeating subunits primarily consisting of glycine (1/3), proline (30%), and hydroxyproline residues.
- Hydroxyproline is a modified form of proline; these modifications occur post-translationally after protein synthesis.
Structural Details
- The arrangement of collagen involves repeating units where glycine precedes proline or hydroxyproline. This sequence contributes to its structural integrity.
- Hydroxylysine residues are also present within collagen's structure, further contributing to its stability.
Helical Structure Formation
- The triple helix formation arises from the specific arrangement of amino acids; glycine often occupies the first position followed by proline or hydroxyproline.
Understanding the Structure of Collagen
The Role of Proline in Helical Structures
- Polyproline forms a double helical structure, resembling a twisted rope, which can be analyzed using the Ramachandran plot. This structure is characterized by proline residues winding around each other to create a right-handed helix.
- The formation of this helical structure is favorable; however, the introduction of another helix must consider stereochemical compatibility.
- For structural stability, glycine must be included as it has a small R group (hydrogen), preventing steric hindrance that would disrupt the helical arrangement formed by proline and hydroxyproline.
Importance of Glycine in Collagen Stability
- To stabilize collagen structures, glycine and hydroxyproline are interspersed among prolines. This separation prevents static hindrance between prolines.
- In diagrams representing collagen's structure, hydrogen bonds and hydrophobic interactions are not always depicted but play crucial roles in maintaining stability.
- A maximum of two consecutive prolines or hydroxyprolines can exist without glycine intervening; otherwise, structural integrity is compromised.
Post-translational Modifications and Their Impact
- Glycine's presence is critical for stabilizing collagen due to its ability to facilitate proper spacing between proline residues.
- Hydroxylation of proline occurs through post-translational modifications facilitated by an enzyme called prolyl hydroxylase.
- Vitamin C acts as a cofactor for prolyl hydroxylase; its absence leads to insufficient hydroxylation and reduced structural integrity in collagen fibers.
Enhancing Hydrogen Bond Formation
- Hydroxylating proline increases potential hydrogen bonding interactions within collagen chains, enhancing their overall stability and interaction degree.
- Increased hydrogen bonding contributes significantly to the triple helical structure's strength and resilience against tensile forces applied during physiological activities.
Structural Characteristics of Collagen
- Each peptide chain in collagen exhibits left-handed helicity while collectively forming a right-handed triple helix, contributing to its tensile strength under stress conditions.
Understanding Collagen Structure and Stability
Helical Arrangement and Structural Stability
- Collagen molecules arrange themselves in a right-handed direction, creating a structurally stable left-handed helix.
- Interactions among three amino acid sequences occur through hydrophobic interactions and hydrogen bonding, crucial for collagen's stability.
- The presence of water disrupts hydrogen bonds between helices, emphasizing the need for a water-free environment to maintain structural integrity.
Hydrogen Bonding Dynamics
- Glycine and proline residues interact via hydrogen bonds; proline often lacks hydroxyl groups necessary for bonding.
- Hydroxyproline modifications enhance the electron-accepting capabilities of amino acids, facilitating more robust hydrogen bonding.
- Water can destabilize helical structures by forming competing hydrogen bonds with glycine, leading to helix separation.
Modifications and Complex Structures
- Rare modifications like hydroxyproline or glucose attachments can be found within collagen but are not common.
- Electron micrographs reveal collagen fibrils' tight arrangement resembling ropes, indicating strong structural integrity.
Role of Lysine in Collagen Formation
- Lysine residues contribute significantly to collagen's complex structure through chemical interactions that stabilize the overall formation.
- Collagen fibrils exhibit alternating dark and light bands due to their organized structure; triple helices stack together compactly.
Enzymatic Reactions Involving Lysine
- Lysin oxidase catalyzes the oxidation of lysine residues, forming aldehyde groups essential for further interactions.
- Aldol condensation mechanisms allow oxidized lysines to interact with other amino acids (e.g., histidine), contributing to collagen's stability.
Understanding Collagen Cross-Linking and Its Implications
The Structure and Function of Collagen
- Discussion on the structural integrity provided by glycine, proline, and hydroxyproline in collagen fibers.
- Introduction to the biosynthetic pathway of cross-linking collagen fibers through interactions between lysines and histidines.
- Explanation of how aging affects collagen's chemical reactivity compared to keratin, leading to increased cross-linking.
Aging and Cross-Linking Effects
- Description of enzymatic interactions that promote cross-linking among collagen residues as cells age.
- Insight into how increased cross-linking stiffens muscle tissue over time, affecting tenderness in meat from older animals.
- Comparison between tender meat from younger animals versus tougher meat from older ones due to collagen changes.
Biological Importance vs. Economic Impact
- Discussion on the dual nature of collagen cross-linking: beneficial for organism integrity but detrimental for meat tenderness.
- Emphasis on the necessity of collagen cross-linking for maintaining muscle function as organisms age.
Structural Arrangements of Collagen
- Overview of different arrangements of collagen fibrils in various tissues (e.g., parallel bundles in tendons).
- Examination of how these arrangements contribute to structural integrity across different body parts like skin and cartilage.
Composition and Applications of Collagen
- Highlight on the amino acid composition (glycine, proline, hydroxyproline), which influences structural properties and functionality.
- Importance of specific arrangements in minimizing light scattering within the cornea for optimal vision.
Collagen Abnormalities and Related Diseases
Understanding Collagen Structure and Its Importance
- Collagen abnormalities arise from disruptions in its structure, often caused by water molecules breaking hydrogen bonds within the collagen chain, leading to separation of collagen fibers.
- Aging contributes to these structural changes, resulting in various diseases such as osteogenesis imperfecta, commonly known as brittle bone disease, characterized by fragile bones prone to fractures.
Types of Diseases Linked to Collagen Abnormalities
- Osteogenesis imperfecta is highlighted as a significant condition where bones become weak and easily fractured due to abnormal collagen structure.
- Ehlers-Danlos syndrome is another condition associated with hyper-extensibility of joints and skin, attributed to high elastin content in tissues that leads to excessive elasticity.