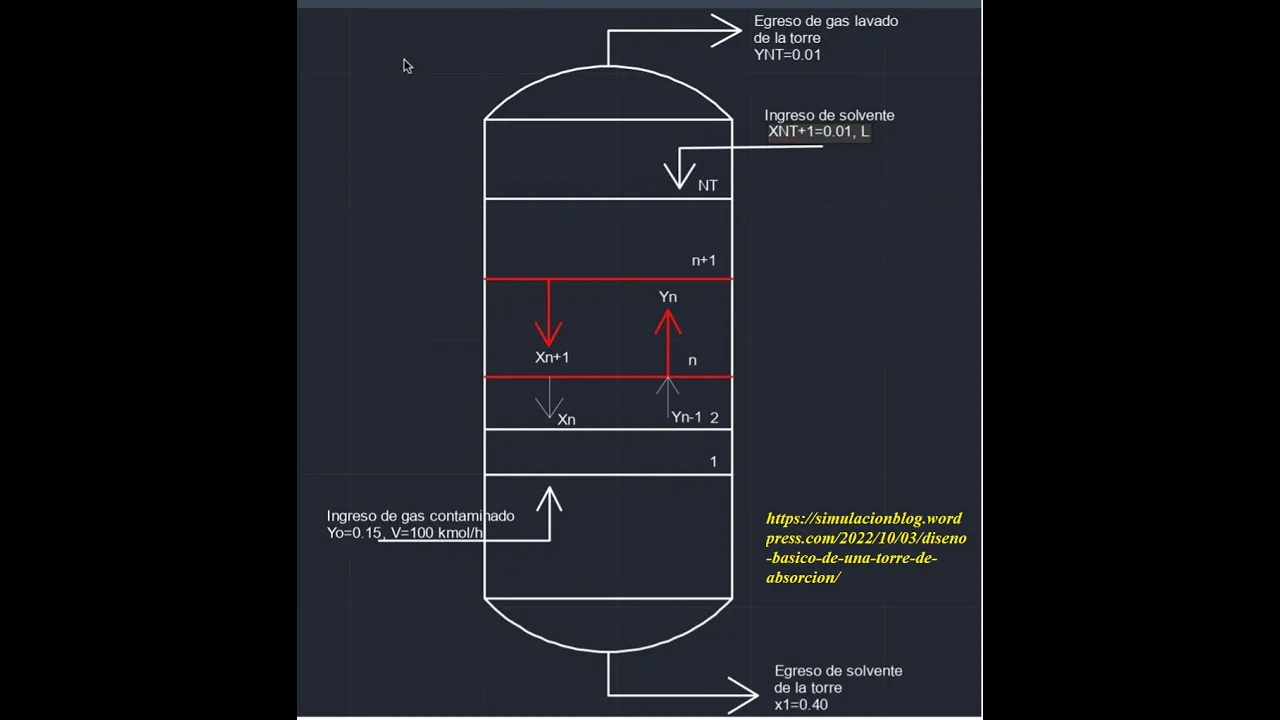
*Columna de absorción: Balance de materia: Problema 2....Parte I
Music
The video starts with music playing.
Understanding the Process of Cleaning Contaminated Air
- The process involves cleaning contaminated air with CO2 by using CO2-free water.
- Operating at 30 degrees Celsius and 1.3 atmospheres, a packed column works in an isothermal manner.
- A mixture of air and SO2 is fed into the base of the column.
- Gas exiting should not exceed 11.7 milligrams of A per liter.
- The system follows Henry's Law, with specific molecular weights provided for various components.
Calculations and Unit Conversions
- Density calculations are based on molecular weights and specific gravities.
- Determining the percentage of SO2 removed involves understanding transferred and untransferred components.
- Homogenizing units is crucial for molar gas phase calculations.
- Calculations involve determining partial pressures using total pressure values.
Conversion to Mass Units
- Converting pressure values to millimeters of mercury aids in further calculations.
- Establishing relationships between densities and fractions simplifies computations.
Chemical Engineering Calculations
In this section, the speaker delves into chemical engineering calculations involving density, molecular weight, and concentration conversions.
Converting Density to Mole
- Density to mass conversion: 2g equals 0.117 kg/m^3.
- Further conversion: 1.828/10^-4 kmol of methanol equals the total density.
Concentration Conversion
- Concentration calculation: The total concentration is determined as 3.495 x 10^-3.
- Relationship between concentrations: Discusses the relationship between different concentrations using various notations like roge2 and cg2.
Calculating Liquid Composition
This part focuses on determining the composition of a liquid mixture leaving a column in terms of weight percentage.
Liquid Composition Calculation
- Initial setup: Setting up the calculation for finding the composition in weight percentage.
- Material balance: Explaining the material balance for a absorption column where concentration is lower at the bottom.
- Solving for x_a1/x_a1: Demonstrates how to solve for x_a1/x_a1 by manipulating equations.
Further Calculations and Formulas
Continuing with calculations involving molecular weights and fractions in chemical engineering contexts.
Molecular Weight Calculation
- Molecular weight computation: Detailed steps on calculating molecular weights for specific substances involved in the process.
- Utilizing values: Using calculated values to determine x_a1/x_a ratio for further analysis.
Fractional Analysis
- Fractional analysis application: Applying fractional analysis to determine liquid phase composition percentages.