2. Sistemas amortiguadores del organismo. Bioquímica.
Understanding Buffer Systems in Biochemistry
Introduction to Buffer Systems
- The concept of biochemical equilibrium is foundational for understanding the body's buffering systems, which help maintain pH levels within physiological limits.
- Physiological buffers present in the human body can capture and release hydrogen ions from both endogenous and exogenous sources.
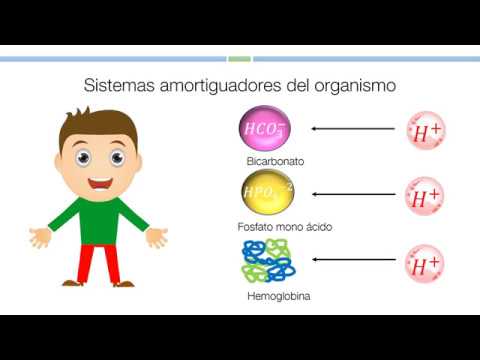
Key Buffers in the Body
- Major buffering agents include bicarbonate, phosphate, hemoglobin, and proteins; these can be found in intracellular or extracellular environments.
- Hemoglobin is highlighted as the most abundant protein in erythrocytes, capable of capturing hydrogen ions produced during carbonic anhydrase reactions.
Mechanism of Intracellular Buffers
- When plasma hydrogen ion concentration increases, they enter cells in exchange for potassium ions, potentially leading to hyperkalemia.
- Conversely, a decrease in plasma hydrogen ions or an increase in bicarbonate results in intracellular buffers releasing hydrogen ions into plasma, which may cause hypokalemia.
Bicarbonate Buffer System
- The bicarbonate buffer system accounts for 75% of physiological pH regulation; it consists of two components: bicarbonate (the metabolic component) and carbon dioxide (the respiratory component).
- The ideal ratio between bicarbonate and carbon dioxide is 20:1 to maintain normal pH levels; any variation disrupts this balance affecting protein ionization.
Functionality of Bicarbonate Buffering
- The primary role of the bicarbonate buffer is to minimize pH changes. An increase in hydrogen ion concentration leads to a decrease in plasma pH.
- Under acidic conditions, bicarbonate neutralizes excess hydrogen ions forming carbonic acid that converts into carbon dioxide and water through a reaction catalyzed by carbonic anhydrase.
Restoration of Normal pH Levels
- When there’s a reduction in hydrogen ion concentration causing increased plasma pH, carbonic acid dissociates back into hydrogen ions and bicarbonate to restore balance.