GCSE Biology - Osmosis
What is Osmosis?
Introduction to Osmosis and Water Concentration
- The video begins by introducing the concept of osmosis, explaining its relevance to water concentration and cellular processes.
- A brief recap of diffusion is provided, defining it as the net movement of particles from an area of higher concentration to one of lower concentration, using oxygen and glucose as examples.
Understanding Osmosis
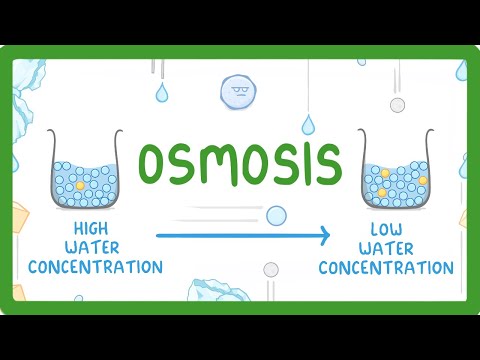
- Osmosis is defined as the net movement of water molecules across a partially permeable membrane from a region of higher water concentration to a region of lower water concentration.
- The term "water concentration" refers to the amount of water compared to solutes (like sugars or salts) dissolved in that water; these solutes are termed "solutes."
Visualizing Water Concentration
- An analogy with two beakers illustrates how solute concentrations affect water concentrations: one beaker has fewer solute particles (higher water concentration), while the other has more (lower water concentration).
- It’s emphasized that it's not just about the volume but rather the proportion of water relative to solutes that determines overall concentration.
Application to Cells
- The video transitions into how osmosis applies within cells, describing a scenario where inside a cell there are many water and some solute particles, while outside there is mostly pure water.
- This setup leads to a situation where water moves from outside (higher concentration) into the cell (lower concentration), illustrating osmosis in action.
Additional Resources