Shell Model of Nucleus
Shell Model Structure of the Nucleus
In this section, the speaker discusses the shell model structure of the nucleus and draws parallels to atomic structure.
Shell Model Similarities with Atomic Structure
- The nucleus's shell model resembles the atomic structure with electrons orbiting around the nucleus in fixed energy levels.
- Complete shell structures corresponding to specific atomic numbers result in stable electronic configurations, akin to inert gases like helium and neon.
Magic Numbers and Stability
- Certain neutron and proton numbers, known as magic numbers (28, 20, 28, 50, 82, 126), lead to highly stable nuclear configurations.
- Nuclei matching these magic numbers exhibit stability, spherical charge distribution, and numerous stable isotopes and isotones.
Double Magic Numbers in Nuclei
This part delves into double magic numbers within nuclei and their significance.
Double Magic Numbers Explanation
- Helium exemplifies a double magic number with two neutrons and protons each.
- Oxygen also showcases double magic numbers with eight protons and neutrons each.
Calcium's Double Magic Numbers
- Calcium features double magic numbers with 20 protons and neutrons each, boasting six stable isotopes.
Stable Isotopes and Isotones
The discussion shifts towards stable isotopes, isotones, and their characteristics.
Stable Isotopes Insights
- Calcium exhibits six distinct stable isotopes due to variations in neutron count while maintaining a constant proton count.
Tin's Stable Isotopes
- Tin's nucleus containing 50 protons demonstrates ten stable isotopes.
Understanding Isotones
Understanding Nuclear Energy Levels
In this section, the discussion revolves around exploring the energy levels of a harmonic oscillator and how they relate to specific numbers. The need for a precise nuclear potential well, different from square wells or harmonic oscillators, is highlighted.
Exploring Energy Levels and Potential Wells
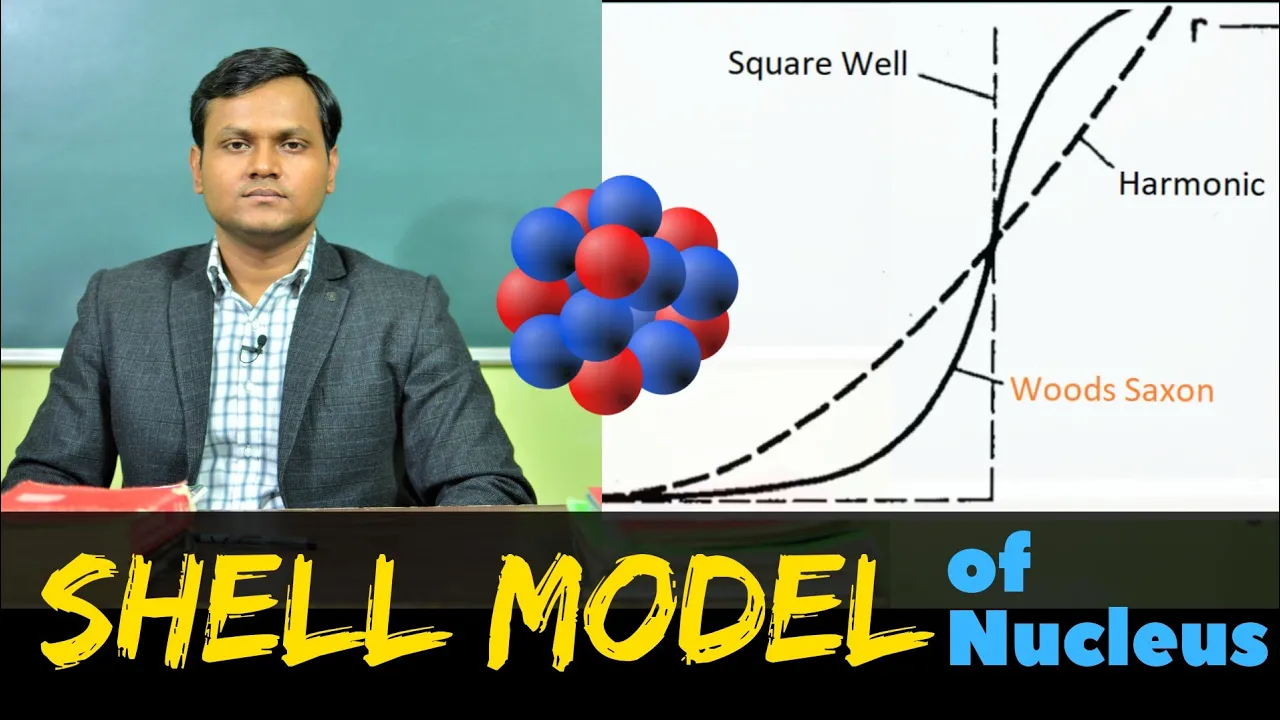
- When solving the quadrupole equation for various nuclear potential wells like infinite square well, finite square well, and harmonic oscillator, the resulting numbers did not align with the expected "magic numbers."
- A more accurate description of a potential well is introduced - the Woods Saxon potential. This potential resembles a square well but with rounded edges, providing a better fit for nuclear systems.
- The Woods Saxon potential was further analyzed using the quadrupole equation, yet it still did not yield the anticipated magic numbers until spin-orbit interaction was considered.
Spin-Orbit Interaction in Atomic Models
- Spin-orbit interaction is explained within atomic models where electrons' orbital motion generates magnetic moments. The interaction between these magnetic moments affects energy levels based on their alignment.
- Electrons possess both orbital and spin-induced magnetic moments that can interact. When aligned similarly, energy increases; when opposite, energy decreases - known as spin-orbit coupling.
- Neutrons and protons exhibit similar spin-orbit interactions due to their magnetic moments and motion inside nuclei. This interaction significantly impacts energy level splitting compared to atomic theory.
Impact of Spin Orbit Interaction on Nuclear Energy Levels
This segment delves into how spin-orbit interactions influence neutron and proton energy levels within nuclei, leading to significant changes in nuclear structure and explaining the concept of magic numbers.
Influence of Spin Orbit Interaction on Energy Levels
- Neutrons and protons experience substantial energy level splitting due to spin-orbit interactions within nuclei. This splitting alters nuclear energy diagrams drastically compared to atomic theory predictions.
- The splitting caused by spin-orbit interactions results in distinct shells forming within nuclei. These shells correspond to specific neutron and proton counts such as 28, 20, 28, 50, 82, and 126 - known as magic numbers.
- By considering spin-orbit interactions alongside Woods Saxon potentials in quadrupole equations, scientists successfully explain magic numbers observed in stable nucleus species through shell structures.
Significance of Shell Model in Nuclear Physics
The discussion emphasizes how the shell model effectively elucidates magic numbers' existence in stable nuclei by integrating Woods Saxon potentials with spin-orbit interactions.
Shell Model Explanation of Magic Numbers
- Through solving quadrupole equations for Woods Saxon potentials while incorporating spin orbit interaction concepts like those proposed by Mayor and Jensen, the shell model provides insights into stable nucleus species' stability.