CLASE TEÓRICA 4 - METABOLOPATÍAS PARTE 1
Metabolopathies and Amyloidosis Overview
Introduction to Metabolopathies
- Dr. Bon Journey introduces the topic of metabolopathies, focusing on arteriosclerosis, diabetes, and gout.
- Arteriosclerosis is categorized into large vessel atherosclerosis, medium vessel arteriolosclerosis, and Monckeberg's calcific sclerosis, with the latter having minimal clinical significance.
Risk Factors for Arteriosclerosis
- Major risk factors include high-fat diets leading to hyperlipidemia, hypertension, diabetes, and long-term tobacco use.
- Minor risk factors consist of obesity linked to sedentary lifestyles and family history; stress also plays a role in metabolic alterations.
Pathogenesis of Arteriosclerosis
- The vascular endothelium undergoes injury that increases permeability and attracts blood cells like monocytes.
- Macrophages formed from monocytes phagocytize lipids and release factors that activate smooth muscle cells in the arterial wall.
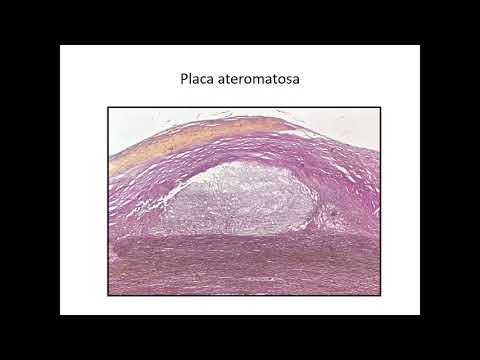
Formation of Atheroma
- LDL cholesterol penetrates the vascular wall contributing to macrophage transformation and promoting further inflammation.
- This process leads to platelet aggregation due to endothelial erosion which can result in complications associated with atheromatous plaques.
Stages of Atheromatous Plaque Development
- Initial lipid accumulation appears as yellow spots on the vascular wall before evolving into raised atheromatous plaques.
- Various risk factors such as hyperlipidemia and hypertension contribute to endothelial injury leading to chronic lesions.
Consequences of Endothelial Injury
- Increased vascular permeability allows leukocyte migration into the arterial wall while stimulating inflammatory responses.
- The presence of LDL cholesterol exacerbates this condition by promoting macrophage activity within the vessel walls.
Structural Changes in Blood Vessels
- Healthy blood vessels maintain laminar flow; however, endothelial damage disrupts this balance leading to turbulent flow conditions.
- Chronic injuries from various risk factors lead to significant changes in vascular structure including increased collagen deposition.
Atherosclerosis and Its Complications
Pathophysiology of Atherosclerosis
- Proliferation of smooth muscle cells migrates from the media to the intima, where macrophages phagocytize lipids bound to LDL, forming lipid streaks and plaques.
- The plaque's core consists of cholesterol-laden macrophages and blood cells, with peripheral proliferation of myointimal cells, smooth muscle cells, and fibroblasts contributing to plaque formation.
- Endothelial alterations such as cracks and erosions promote platelet aggregation; this disrupts hemodynamic forces leading to turbulent flow in the vessel lumen.
- Inflammation within atheromatous plaques involves neovascularization, lymphocytes, macrophages, and extracellular matrix remodeling.
Evolution and Complications of Atheromatous Plaques
- Atheromatous plaques are dynamic structures that can undergo various evolutionary processes including ulceration and intraplaque hemorrhaging.
- An aneurysm represents vascular dilation due to weakened walls; hypertension exacerbates this condition by increasing intravascular pressure leading to potential rupture.
- Thrombus generation is influenced by endothelial injury (Birch's triad), increased peripheral resistance, and turbulent flow promoting platelet aggregation.
Clinical Implications of Atherosclerosis
- Total occlusion or partial narrowing from thrombus growth leads to ischemic phenomena such as infarction or necrosis in affected tissues.
- Fragments from thrombi may travel through circulation causing embolic events in distant organs resulting in significant complications like organ infarcts.
Macroscopic Observations
- Macroscopic images reveal arterial wall lesions characterized by yellowish lipid streaking versus raised atheromatous plaques indicating severe endothelial alteration.
- The aorta exhibits extensive erosion with a "cobblestone" appearance due to multiple atheromatous plaques alongside areas of hemorrhage or dystrophic calcification.
Common Locations for Atherosclerosis
- Most frequent sites include abdominal aorta, iliac arteries, coronary arteries, thoracic aorta, femoral arteries, carotid arteries (internal/external), vertebral arteries, and middle cerebral artery supplying the brain.
Consequences on Vital Organs
- Heart-related complications include chronic ischemic heart disease manifesting as angina pectoris or myocardial infarction due to terminal circulation issues in the myocardium.
Understanding Cerebral Microcirculation Disorders and Diabetes
Cerebral Microcirculation and Its Impact on Health
- The discussion begins with the relationship between dementia and disorders in cerebral microcirculation, highlighting how renal insufficiency and renal infarction contribute to these conditions.
- It is noted that hyaline and hyperplastic arteriosclerosis affect medium and small caliber arteries, characterized by vascular wall thickening leading to reduced blood flow and ischemia.
- Chronic ischemia can lead to benign hypertension, while hyperplastic arteriosclerosis presents a more aggressive behavior with serious complications due to concentric laminated thickening of the vascular wall.
- Hyperplasia of myointimal cells, fibrin deposits, and necrosis are discussed as consequences of vascular changes, resulting in marked stenosis and malignant hypertension.
- A comparison is made between benign arteriolosclerosis (hyaline thickening) and hyperplastic arteriolosclerosis (onion-skin appearance), emphasizing differences in cellular proliferation.
Pathophysiology of Diabetes Mellitus
- Transitioning to diabetes mellitus, it is defined by elevated blood glucose levels (>120 mg/dL), which may be primary or secondary due to other conditions like glucocorticoid use or endocrine tumors.
- Primary diabetes is emphasized, particularly Type 1 (insulin-dependent) and Type 2 (insulin-resistant), each having distinct metabolic implications for carbohydrates, lipids, and proteins.
- Type 1 diabetes involves genetic factors linked to chromosome 6 affecting autoimmune responses that destroy insulin-secreting pancreatic beta cells.
- Environmental factors such as viral exposure or toxins also play a role in Type 1 diabetes pathogenesis alongside genetic predispositions.
- In contrast, Type 2 diabetes involves autosomal dominant genetic influences leading to insulin resistance primarily due to mutations affecting insulin release mechanisms.
Metabolic Consequences of Diabetes
- Ketoacidosis arises from lipid metabolism alterations causing increased circulating free fatty acids; this leads to dehydration at the cellular level along with metabolic acidosis.
- The discussion highlights how advanced glycation end products form during lipid metabolism disturbances contributing further to cellular damage within diabetic patients.
Understanding Diabetes: Type 1 and Type 2
Pathophysiology of Type 1 Diabetes
- In type 1 diabetes, there is a significant decrease in insulin secretion due to an autoimmune condition affecting the pancreatic beta cells, leading to insulitis characterized by lymphocytic infiltration and fibrosis.
- The histological examination reveals extensive inflammatory lymphocytic infiltrate in the pancreas, resulting in destruction of beta cells along with atrophy and fibrosis.
Pathophysiology of Type 2 Diabetes
- In contrast to type 1, type 2 diabetes presents with normal or increased numbers of pancreatic beta cells; however, there is a resistance to insulin action in peripheral tissues due to receptor alterations.