Ciclo de Rankine | Termodinâmica
New Section
In this section, the speaker introduces the topic of thermodynamics and specifically focuses on the Rankine cycle.
Introduction to Thermodynamics and Rankine Cycle
- The discussion begins with an overview of thermodynamic diagrams and the basic components of the Rankine cycle.
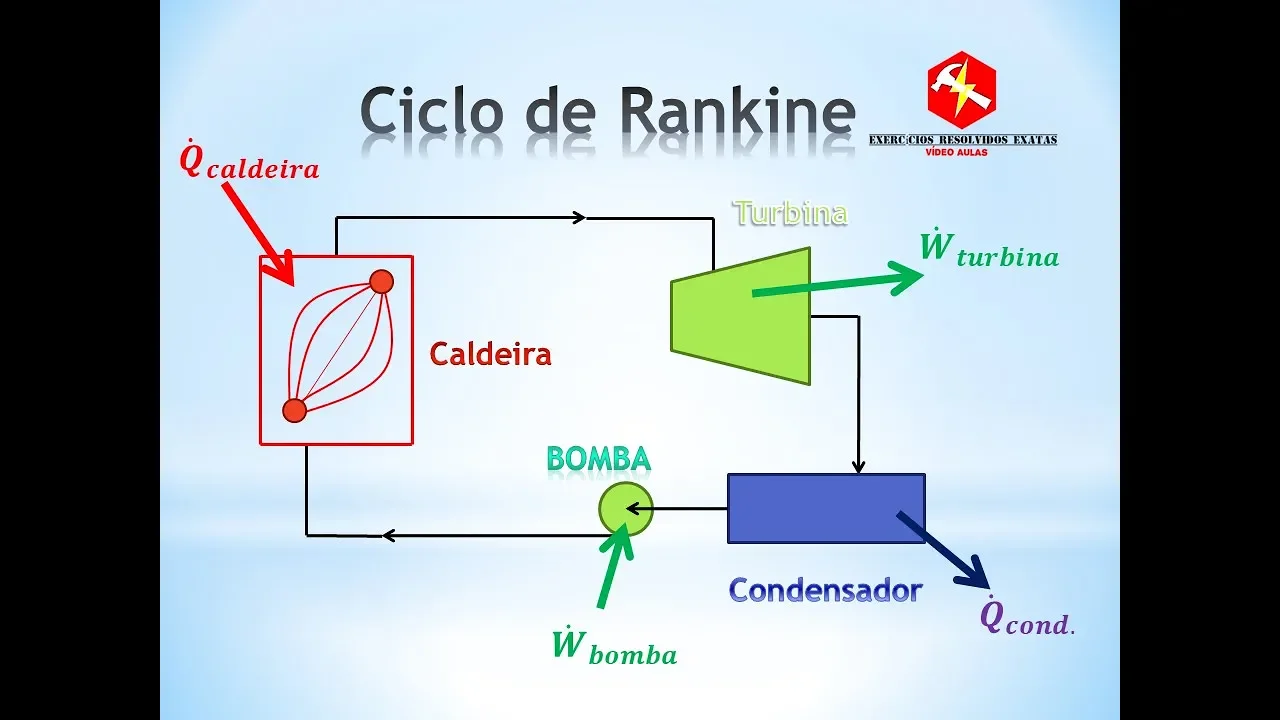
- Explains the simple Rankine cycle, highlighting its four main components and emphasizing the calculation of heat exchanges and work interactions within the cycle.
- Emphasizes understanding heat exchanges, work interactions, and calculating the efficiency of a Rankine cycle based on heat calculations.
- Teases upcoming content by mentioning examples of exercises related to Rankine cycles that will be covered in subsequent videos.
- Explores thermodynamic diagrams further, focusing on how they relate two properties of a fluid, typically water, through temperature and entropy.
Understanding Thermodynamic Diagrams
This section delves into the details of thermodynamic diagrams, particularly focusing on pressure lines and phase changes within these diagrams.
Thermodynamic Diagram Details
- Discusses how thermodynamic diagrams relate properties like temperature and entropy for fluids such as water.
- Highlights pressure lines' significance in indicating constant pressure regions during phase changes within processes.
- Explains regions within the diagram representing liquid subcooled, saturated liquid-vapor mixture, and superheated vapor states based on temperature variations.
- Describes transitions between liquid subcooled, saturated liquid-vapor mixture, and superheated vapor states as heat is added to or removed from a system.
- Discusses energy differences between different states in a Rankine cycle system concerning saturated vapor, saturated liquid-vapor mixture, and liquid subcooled phases.
Components of Simple Rankine Cycle
This part focuses on detailing each component of a simple Rankine cycle - boiler, turbine, condenser, and pump - along with their functions in the overall process.
Components Explanation
Energy Conversion Process in a Rankine Cycle
In this section, the speaker explains the energy conversion process within a Rankine cycle, focusing on the transformation of heat into work to generate electricity.
Energy Production in Turbine
- The primary goal is to produce work in the turbine to convert it into electrical energy.
- Heat is added to the fluid in the boiler to increase its temperature before entering the turbine.
- The condenser removes heat from the fluid, transitioning it from a liquid-vapor state back to liquid form.
Fluid States and Functions of Components
- The pump increases fluid pressure by adding work, preparing it for re-entry into the boiler.
- The fluid transitions from liquid to vapor in the boiler due to added heat.
Understanding Turbine Operation and Efficiency
This section delves into the operation of turbines within a Rankine cycle, emphasizing ideal conditions without energy or pressure losses.
Turbine State and Expansion Process
- Fluid entering the turbine is at high pressure and superheated vapor state (Point 1).
- During expansion in the turbine (Point 2), pressure decreases as energy is extracted from the fluid.
Entropy and Irreversibility
- Pressure reduction leads to reaching a saturated vapor state (liquid-vapor mix), with constant entropy change indicating an irreversible process.
Condenser Functionality and Heat Removal
Exploring how condensers operate within a Rankine cycle by extracting heat from fluids.
Heat Extraction Process
- Condenser removes heat, converting vapor back into liquid form (Point 3).
Detailed Thermodynamics Analysis
In this section, the speaker delves into a detailed thermodynamic analysis, focusing on calculations involving turbines and heat exchange processes in a steady-state regime.
Calculations for Turbine Processes
- At point 1, values of temperature, pressure, and entropy are determined for superheated vapor state.
- Transitioning from point 1 to point 2 involves considering temperature, pressure, and specific volume changes due to vapor-liquid mixture.
- Entropy equivalence between states 2 and 1 is crucial for determining properties during phase change.
Heat Exchange in Condenser
- The condenser process involves extracting heat energy as the fluid transitions from vapor to liquid state at point 3.
- Point 3 signifies complete liquid state with zero quality and defined entropy and specific volume values.
Pressure Considerations in Condenser and Boiler
- Pressure equality between points 1.3 and 2 ensures constant pressure during condensation process.
- Pressure remains constant from boiler (point 4) to condenser (point 3), aiding in simplifying calculations.
Entropy Calculation Methods
This segment explores entropy calculation methodologies, particularly focusing on pump processes and their impact on system efficiency.
Pump Process Simplifications
- Analyzing entropy variation in pumps involves treating the process as isentropic due to its incomprehensible nature.
- Entropy change equations simplify by considering constant volume conditions for fluid passing through pump stages.
Application of First Law of Thermodynamics
The application of the first law of thermodynamics is discussed concerning heat exchange processes within components like boilers and turbines.
First Law Application
- Heat exchange calculations involve utilizing enthalpy differences across components like boilers (h1-h4).
Calculating Efficiency of a System
In this section, the speaker explains how to calculate the efficiency of a system based on the values obtained for work and heat.
Calculating Total Efficiency
- The total efficiency of a cycle is determined by dividing the net work done by the system by the heat input into the system.
- : Net work is calculated as the work done by the turbine minus the work done by the pump.
- Useful work is what can be utilized after considering all energy expenditures, such as electricity used by pumps.
- : It involves subtracting the pump's work from the turbine's work.
- To calculate specific components' works, such as turbine and pump:
- : Turbine's work = h1 - h2
- : Pump's work = h4 - h3
- A future exercise will involve determining efficiencies based on specified states and calculations.
- : This exercise will focus on calculating system efficiency.
Upcoming Topics in Thermodynamics
The speaker hints at future topics to be covered in upcoming classes related to thermodynamics.
Future Discussions
- In subsequent classes, topics like reheat cycles and regenerative cycles will be explored further.
- Exercises in these classes will involve determining efficiencies for various cases and states.