How does a hydrogen fuel cell work? (AKIO TV)
Introduction to Hydrogen Fuel Cells
In this section, the speaker introduces hydrogen gas (H2) as a fuel source and explains its potential as a clean energy option. The concept of using hydrogen fuel cells to power machines and vehicles is also introduced.
How Hydrogen Fuel Cells Work
- A fuel cell is used to convert hydrogen and oxygen into electricity.
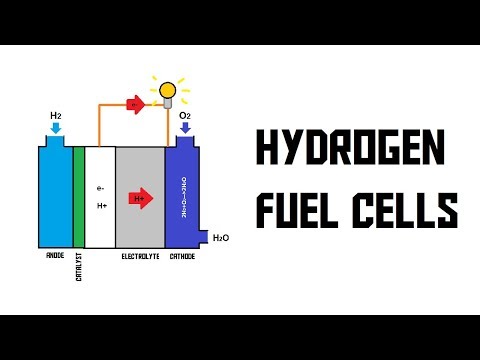
- A schematic drawing of a hydrogen fuel cell is shown, with an anode on the left side and a cathode on the right side.
- Inside the fuel cell, there is a catalyst and an electrolyte between the anode and cathode.
- When the system is turned on, hydrogen in the anode wants to react with oxygen in the cathode to form water.
- However, the electrolyte prevents neutral hydrogen atoms from passing through it.
- The catalyst splits off electrons from protons in the hydrogen atoms, allowing protons to pass through the electrolyte.
- To complete the reaction, electrons are transported via a wire, creating an electric current that can be used as power.
- The combination of protons meeting with oxygen forms water as waste product.
Drawbacks of Fuel Cells
- Fuel cells can get hot during operation and require proper cooling to prevent damage.
- Fuel cells are expensive, primarily due to the cost of the catalyst used in the process.
Conclusion
Hydrogen fuel cells offer a clean and efficient way to generate electricity by utilizing hydrogen and oxygen. The process involves a catalyst, an electrolyte, and the transportation of electrons to create an electric current. However, fuel cells have drawbacks such as heat generation and high costs associated with catalyst usage.